Chemiluminescence Immunoassay Market - Global Forecast to 2031
化学発光免疫測定市場 - 製品(機器、消耗品)、技術(CLEIA、ECLI、微粒子CLIA)、サンプルタイプ(血液、唾液)、用途(腫瘍学、心臓病学)、エンドユーザー(病院、臨床検査室) - 2031年までの世界予測
Chemiluminescence Immunoassay Market by Product (Instruments, Consumables), Technology (CLEIA, ECLI, Microparticle CLIA), Sample Type (Blood, Saliva), Application (Oncology, Cardiology), End User (Hospital, Clinical Laboratory) - Global Forecast to 2031
| 出版 | MarketsandMarkets |
| 出版年月 | 2026年03月 |
| ページ数 | 383 |
| 図表数 | 348 |
| 価格 | 記載以外のライセンスについてはお問合せください |
| シングルユーザ | USD 4,950 |
| 種別 | 英文調査報告書 |
| 商品番号 | SMR-1524115241 |
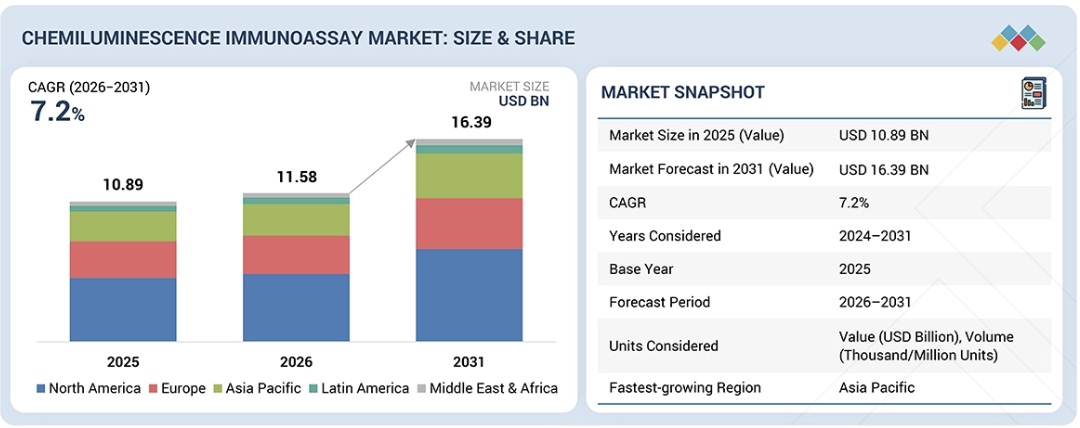
化学発光免疫測定市場は、2026年の115億8,000万米ドルから2031年には163億9,000万米ドルに達し、2026年から2031年にかけて7.2%のCAGRで成長すると予測されています。
がん、心血管疾患、甲状腺疾患、ウイルス感染など、慢性疾患や感染症を特定するための正確な診断検査の必要性が高まる中、CLIA検査システムの世界的な市場は拡大しています。早期疾患発見のニーズの高まりと予防医療への取り組みにより、CLIA検査は優れた感度と幅広い検査範囲を備え、微量のバイオマーカーを特定できるため、その利用が拡大しています。自動化されたハイスループット検査システムの開発、より安定した検査材料、複数の検査を同時に実施する能力により、検査室の生産性が向上し、検査時間が短縮されました。市場の成長は、医療施設の改善、検査室システムの拡張、そして新興市場における医療費の増加に支えられています。個別化医療、医薬品開発、臨床研究におけるバイオマーカー検査の重要性の高まり、そして厳格な血液スクリーニング規制と健康診断の意識の高まりにより、世界中の病院や検査室におけるCLIAシステムのニーズが高まっています。
調査範囲:
本レポートは、CLIA市場を分析し、製品タイプ、サンプルタイプ、技術、用途、エンドユーザーといった様々なセグメントに基づいて、市場規模と将来の成長可能性を推定することを目的としています。また、市場で入手可能な様々なCLIAの製品ポートフォリオマトリックスも掲載しています。さらに、主要プレーヤーの競合分析に加え、企業概要、製品ラインナップ、主要な市場戦略も提供しています。
本レポートを購入する理由
本レポートは、市場リーダー企業や新規参入企業にとって、CLIA市場全体および各サブセグメントの収益予測に関する情報を提供します。本レポートは、関係者が競争環境を理解し、ビジネスポジショニングを改善し、適切な市場開拓戦略を策定するための洞察を深めるのに役立ちます。さらに、本レポートは、関係者が市場の動向を把握し、主要な市場牽引要因、制約要因、課題、機会に関する情報を提供するのに役立ちます。
本レポートは、以下の点について洞察を提供します。
- 主要な推進要因(世界的な慢性疾患および感染症の発生率の増加、近年のCLIA技術の進歩、世界的な高齢化人口の急増、バイオテクノロジーおよびバイオ医薬品産業の成長)、制約要因(CLIAの設置と維持に伴う高コスト、製品リコールおよび故障)、課題(熟練した専門家の不足、CLIAに関連するリスク)、機会(新興国における成長の可能性)の分析
- 製品強化/イノベーション:世界のCLIA市場における新製品の発売と予想されるトレンドに関する包括的な詳細情報
- 製品開発/イノベーション:世界のCLIA市場における今後のトレンド、研究開発活動、新製品の発売に関する詳細な洞察
- 市場開発:製品タイプ、技術、サンプルタイプ、アプリケーション、エンドユーザー別に、魅力的な新興市場に関する包括的な情報
- 市場の多様化:新製品やサービス、または製品やサービスの強化、成長地域、最近の世界のCLIA市場における開発および投資
- 競合評価:世界のCLIA市場における主要競合他社の市場シェア、成長計画、製品・サービスの提供、および能力の徹底的な評価
Report Description
The chemiluminescence immunoassay market is projected to reach USD 16.39 billion by 2031 from USD 11.58 billion in 2026, at a CAGR of 7.2% from 2026 to 2031.
Chemiluminescence Immunoassay Market – Global Forecast to 2031
The global market for CLIA testing systems is growing as people need accurate diagnostic tests to identify chronic and infectious diseases, including cancer, cardiovascular disorders, thyroid diseases, and viral infections. The increasing need for early disease detection, together with preventive healthcare approaches, leads to greater use of CLIA testing because it provides excellent sensitivity and a wide testing range and can identify trace amounts of biomarkers. The development of automated high-throughput testing systems, together with more stable testing materials and the ability to conduct multiple tests simultaneously, has improved laboratory productivity while decreasing testing duration. The market growth benefits from improved healthcare facilities, expanded testing laboratory systems, and increased medical expenditures that emerging markets are experiencing. The rising importance of biomarker testing for personalized medicine, drug development, and clinical research, together with strict blood screening regulations and health checkup awareness, boosts the need for CLIA systems in hospitals and reference laboratories throughout the world.
“Based on the product, the consumables segment is expected to grow at the highest CAGR in the CLIA market.”
The consumables segment of the CLIA market is growing because diagnostic testing requires laboratories to use reagents, calibrators, controls, and reaction vessels multiple times throughout their workday. Assay kits and reagent packs that hospitals and reference laboratories use daily see increased demand because infectious disease tests, oncology marker tests, cardiac biomarker tests, endocrine disorder tests, and therapeutic drug monitoring tests are being performed more frequently. Consumables require continuous replacement, which generates ongoing revenue while driving market growth, in contrast to analyzers. The adoption of new technologies that enhance reagent stability and ready-to-use product development and extended shelf-life capabilities leads to higher operational efficiency and lower material waste in laboratory workflows. The expansion of automated high-throughput CLIA systems, together with increased preventive health screening programs, higher blood safety testing, and expanded test menu options, is driving up consumables consumption, which makes this segment a key driver of overall CLIA market expansion.
“Based on application, the oncology segment is expected to grow with the highest CAGR in the CLIA market.”
The CLIA market in oncology applications shows growth because cancer cases are increasing worldwide, and there is greater demand for early diagnosis, prognosis evaluation, and treatment assessment. CLIA technology enables the detection of tumor markers through highly sensitive and specific methods, which include PSA, CA-125, CEA, AFP, and HER2 as diagnostic tools for initial patient evaluation and progress assessment of their medical condition. The rise in biomarker-based testing and precision oncology practices has created a need for dependable immunoassays, which assist in determining targeted therapies and individualized treatment options. The clinical use of CLIA tests has grown because these tests now help doctors monitor patient treatment response, identify cancer recurrences, and assess patients who have a high risk of developing cancer. The introduction of better assay systems, which enhance test sensitivity through automated processes and multiplex testing features, will continue to drive businesses toward adopting CLIA systems in their cancer diagnostic processes.
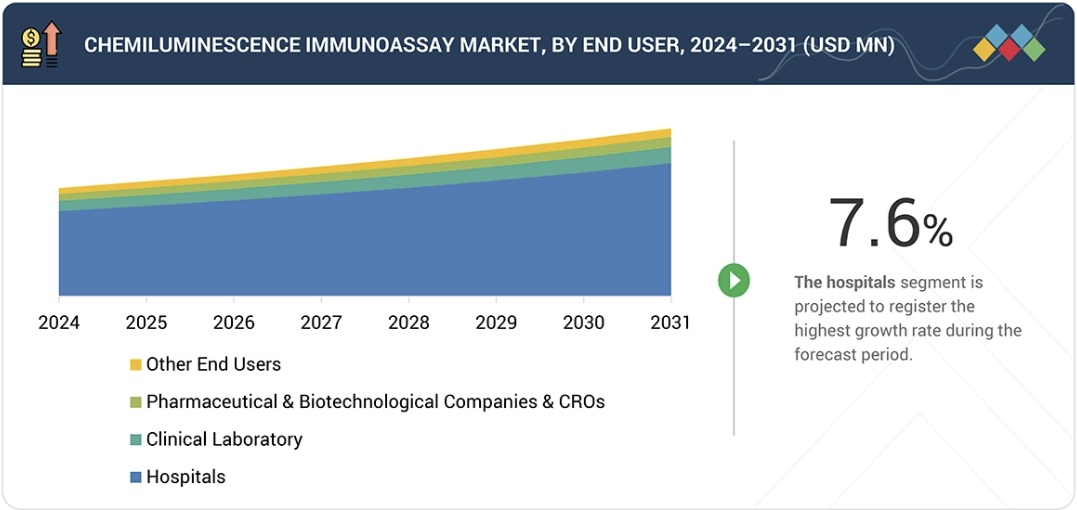
“Based on the end user, the hospitals segment is expected to grow by the highest CAGR in the CLIA market.”
Hospitals need to conduct accurate tests for various medical conditions, including cardiac markers, sepsis indicators, cancer biomarkers, hormone levels, and infectious pathogens. The CLIA systems enable emergency departments, intensive care units, and inpatient services to start treatment without delay through their advanced sensitivity, extensive testing capabilities, and quick result delivery. The move to centralized diagnostic systems, together with automated diagnostic machines, helps hospitals to cut down on manual tasks while achieving better operational results through standardized processes. The worldwide implementation of CLIA analyzers and assays in hospital systems accelerates because hospitals expand their facilities, and preventive health screening programs become more popular, accreditation and quality standards increase, and hospitals need complete laboratory information systems.

Chemiluminescence Immunoassay Market – Global Forecast to 2031 – region
“APAC is estimated to register the highest CAGR during the forecast period.”
The CLIA market in the Asia Pacific region is growing due to three factors: expanding healthcare, rising healthcare spending, and improved distribution of diagnostic services in countries such as China, India, and Southeast Asia. The rising need for precise early diagnostic tests results from the increasing incidence of chronic illnesses, infectious diseases, and conditions linked to lifestyle choices. The government modernization projects, which include hospital upgrades, laboratory automation, and the development of a national screening program, accelerate the implementation of high-throughput CLIA systems. The demand for advanced immunoassay technologies grows due to three factors, which include increasing public knowledge about preventive healthcare, expanding private diagnostic laboratory networks, and rising medical tourism. The regional growth of the market benefits from three factors, which include a large patient base, better insurance access, and the market’s shift toward automated laboratory systems that manage high testing loads. The widespread implementation of CLIA platforms throughout APAC results from two factors, which include ongoing international diagnostic company expansion and the rise of affordable domestic manufacturers.
Key players in the CLIA market
The key players in the market include F. Hoffmann-La Roche Ltd. (Switzerland), Abbott Laboratories (US), Siemens Healthineers (Germany), Danaher Corporation (US), DiaSorin S.p.A. (Italy), QuidelOrtho Corporation (US), Sysmex Corporation (Japan), Shenzhen New Industry Biomedical Engineering Co., Ltd. (SNIBE Diagnostics) (China), Tosoh Corporation (Japan), Werfen (Spain), Bio-Rad Laboratories, Inc. (US), Shenzhen Mindray Biomedical Electronics Co., Ltd. (China), Fujirebio (Japan), HUMAN Gesellschaft für Biochemica und Diagnostica mbH. (Germany), EUROIMMUN Medizinische Labordiagnostika AG (Germany), Agappe Diagnostics Ltd (India), Zecen Biotech Co., Ltd (China), Maccura Biotechnology Co., Ltd (China), Autobio Diagnostics Co., Ltd. (China), Artron Laboratories Inc.(Canada), Abnova Corporation (Taiwan), Beijing Hotgen Biotech Co., Ltd (China), and Shanghai Kehua Bio-Engineering Co., Ltd. (China).

Chemiluminescence Immunoassay Market – Global Forecast to 2031 – ecosystem
Research Coverage:
The report analyzes the CLIA market and aims at estimating the market size and future growth potential of this market based on various segments such as product type, sample type, technology, application, and end user. The report also includes a product portfolio matrix of various CLIA available in the market. The report also provides a competitive analysis of the key players in this market, along with their company profiles, product offerings, and key market strategies.
Reasons to Buy the Report
The report will help the market leaders/new entrants in this market with information on the closest approximations of the revenue numbers for the overall CLIA market and the subsegments. This report will help stakeholders understand the competitive landscape and gain more insights to position their businesses better and plan suitable go-to-market strategies. The report also helps stakeholders understand the pulse of the market and provides them with information on key market drivers, restraints, challenges, and opportunities.
This report provides insights into the following pointers:
- Analysis of key drivers (increasing incidences of chronic and infectious diseases globally, advancements in improving CLIA technologies in recent years, rapid increase in geriatric population globally, growth of biotechnology and biopharmaceutical industries), restraints (high cost associated with placement and maintenance of CLIA, product recalls and failures), challenges (shortage of skilled professionals, risks associated with CLIAs), and opportunities (growth potential in emerging economies)
- Product Enhancement/Innovation: Comprehensive details about new product launches and anticipated trends in the global CLIA market
- Product Development/Innovation: Detailed insights on upcoming trends, research & development activities, and new product launches in the global CLIA market
- Market Development: Comprehensive information on the lucrative emerging markets by product type, technology, sample type, application, and end user
- Market Diversification: Exhaustive information about new products and services or product and service enhancements, growing geographies, recent developments, and investments in the global CLIA market
- Competitive Assessment: Thorough evaluation of the market shares, growth plans, offerings of products and services, and capacities of the major competitors in the global CLIA market
Table of Contents
1 INTRODUCTION 30
1.1 STUDY OBJECTIVES 30
1.2 MARKET DEFINITION 30
1.3 STUDY SCOPE 31
1.3.1 MARKETS COVERED & REGIONAL SCOPE 31
1.3.2 INCLUSIONS & EXCLUSIONS 32
1.3.3 YEARS CONSIDERED 32
1.3.4 CURRENCY CONSIDERED 33
1.3.5 UNITS CONSIDERED 33
1.4 STAKEHOLDERS 33
1.5 SUMMARY OF CHANGES 34
2 EXECUTIVE SUMMARY 35
2.1 MARKET HIGHLIGHTS & KEY INSIGHTS 35
2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS 36
2.3 DISRUPTIVE TRENDS IN CHEMILUMINESCENCE IMMUNOASSAY (CLIA) MARKET 37
2.4 HIGH-GROWTH SEGMENTS 38
2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST 39
3 PREMIUM INSIGHTS 41
3.1 CHEMILUMINESCENCE IMMUNOASSAY OVERVIEW 41
3.2 ASIA PACIFIC CHEMILUMINESCENCE IMMUNOASSAY, BY PRODUCT AND COUNTRY 42
3.3 CHEMILUMINESCENCE IMMUNOASSAY: DEVELOPED MARKETS VS.
EMERGING ECONOMIES 43
3.4 CHEMILUMINESCENCE IMMUNOASSAY: GEOGRAPHIC GROWTH OPPORTUNITIES 44
3.5 CHEMILUMINESCENCE IMMUNOASSAY: REGIONAL MIX 45
4 MARKET OVERVIEW 46
4.1 INTRODUCTION 46
4.2 MARKET DYNAMICS 47
4.2.1 DRIVERS 47
4.2.1.1 Increasing incidence of chronic and infectious diseases globally 47
4.2.1.2 Advancements in chemiluminescence immunoassay technologies in recent years 49
4.2.1.3 Rapid increase in geriatric population globally 50
4.2.1.4 Growth of biotechnology and biopharmaceutical industries 51
4.2.2 RESTRAINTS 52
4.2.2.1 High cost of chemiluminescence systems and reagents 52
4.2.2.2 Lack of regular quality control procedures for monitoring and detecting cross-reactivity and interference 52
4.2.3 OPPORTUNITIES 53
4.2.3.1 High growth prospects for players in emerging economies 53
4.2.3.2 Increasing number of collaborations and partnerships 54
4.2.4 CHALLENGES 54
4.2.4.1 Unfavorable reimbursement scenario and budgetary constraints in healthcare systems 54
4.2.4.2 Lack of skilled professionals and aging workforce 55
4.3 UNMET NEEDS & WHITE SPACES 55
4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES 57
4.4.1 INTERCONNECTED MARKETS 57
4.4.2 CROSS-SECTOR OPPORTUNITIES 58
4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS 58
5 INDUSTRY TRENDS 60
5.1 PORTER’S FIVE FORCES ANALYSIS 60
5.1.1 THREAT OF NEW ENTRANTS 61
5.1.2 THREAT OF SUBSTITUTES 61
5.1.3 BARGAINING POWER OF SUPPLIERS 62
5.1.4 BARGAINING POWER OF BUYERS 62
5.1.5 INTENSITY OF COMPETITIVE RIVALRY 62
5.2 MACROECONOMIC INDICATORS 62
5.2.1 INTRODUCTION 62
5.2.2 GDP TRENDS & FORECAST 62
5.2.3 TRENDS IN GLOBAL HEALTHCARE INDUSTRY 64
5.2.4 TRENDS IN GLOBAL MEDICAL DEVICE INDUSTRY 65
5.3 VALUE CHAIN ANALYSIS 66
5.4 SUPPLY CHAIN ANALYSIS 67
5.5 ECOSYSTEM ANALYSIS 68
5.6 PRICING ANALYSIS 70
5.6.1 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY KEY PLAYER, 2023–2025 (USD) 71
5.6.2 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY REGION, 2023–2025 (USD) 71
5.6.3 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY TECHNOLOGY, 2023–2025 (USD) 73
5.6.4 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY CONSUMABLES (KITS, REAGENTS, AND OTHERS), BY SAMPLE TYPE,
2023–2025 (USD) 73
5.7 TRADE ANALYSIS 73
5.7.1 IMPORT DATA FOR HS CODE 9027.89, 2021–2025 74
5.7.2 EXPORT DATA FOR HS CODE 9027.89, 2021–2025 74
5.8 KEY CONFERENCES & EVENTS, 2026–2027 75
5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES 76
5.10 INVESTMENT & FUNDING SCENARIO 77
5.11 CASE STUDY ANALYSIS 78
5.11.1 CASE STUDY 1: ROCHE COBAS CLIA DRIVES HIGH-VOLUME TESTING PERFORMANCE 78
5.11.2 CASE STUDY 2: ABBOTT ARCHITECT DRIVES REGIONAL
MULTI-CENTER LAB EXPANSION 78
5.11.3 CASE STUDY 3: SIEMENS ADVIA CENTAUR XP BOOSTS
GOVERNMENT LAB TESTING EFFICIENCY 79
5.12 IMPACT OF 2025 US TARIFFS ON CHEMILUMINESCENCE IMMUNOASSAY MARKET 79
5.12.1 INTRODUCTION 79
5.12.2 KEY TARIFF RATES 80
5.12.3 PRICE IMPACT ANALYSIS 81
5.12.4 IMPACT ON COUNTRIES/REGIONS 81
5.12.4.1 US 81
5.12.4.2 Europe 81
5.12.4.3 Asia Pacific 82
5.12.5 IMPACT ON END-USE INDUSTRIES 82
5.12.5.1 Hospitals 82
5.12.5.2 Clinical laboratories 82
5.12.5.3 Pharmaceutical & biotechnology companies and contract research organizations 83
6 STRATEGIC DISRUPTIONS THROUGH TECHNOLOGY, PATENTS, AND
DIGITAL & AI ADOPTION 84
6.1 TECHNOLOGY ANALYSIS 84
6.1.1 KEY EMERGING TECHNOLOGIES 84
6.1.1.1 Nano-engineered chemiluminescent labels 84
6.1.1.2 Digital immunoassay platforms 84
6.1.2 COMPLEMENTARY TECHNOLOGIES 84
6.1.2.1 Bead-based & microarray chemiluminescence 84
6.1.2.2 Integration with microfluidic platforms 85
6.1.3 ADJACENT TECHNOLOGIES 85
6.1.3.1 Enzyme-linked immunosorbent assay (ELISA) 85
6.2 TECHNOLOGY/PRODUCT ROADMAP 85
6.3 PATENT ANALYSIS 86
6.3.1 INSIGHTS ON PATENT PUBLICATION TRENDS, TOP APPLICANTS, AND JURISDICTION FOR CHEMILUMINESCENCE IMMUNOASSAY MARKET, JANUARY 2015–SEPTEMBER 2025 86
6.3.2 LIST OF MAJOR PATENTS, 2022–2025 88
6.4 FUTURE APPLICATIONS 89
6.4.1 PERSONALIZED & PRECISION MEDICINE 89
6.4.2 REMOTE MONITORING & DECENTRALIZED TESTING 89
6.4.3 PREDICTIVE DIAGNOSTICS & CLINICAL DECISION SUPPORT 89
6.4.4 INTEGRATION WITH DIGITAL HEALTH ECOSYSTEMS 89
6.4.5 NEXT-GENERATION BIOMARKER DISCOVERY & MULTIPLEX TESTING 90
6.5 IMPACT OF GEN AI ON CHEMILUMINESCENCE IMMUNOASSAY MARKET 90
6.5.1 IMPACT OF AI/GEN AI ON INTERCONNECTED & ADJACENT ECOSYSTEMS 92
6.6 SUCCESS STORIES AND REAL-WORLD APPLICATIONS 92
6.6.1 IMPROVED OUTCOMES IN CRITICAL CARE & EMERGENCY DIAGNOSTICS 92
6.6.2 PUBLIC HEALTH SCREENING & INFECTIOUS DISEASE CONTROL 92
6.6.3 ENDOCRINOLOGY & CHRONIC DISEASE MANAGEMENT 93
6.6.4 ONCOLOGY DIAGNOSTICS & THERAPY MONITORING 93
6.6.5 MATERNAL & NEONATAL SCREENING PROGRAMS 93
6.6.6 DECENTRALIZED & MID-VOLUME LABORATORY ADOPTION 93
6.6.7 REAL-WORLD IMPACT 93
7 SUSTAINABILITY & REGULATORY LANDSCAPE 94
7.1 REGULATORY LANDSCAPE 94
7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 94
7.1.2 INDUSTRY STANDARDS 97
7.1.2.1 ISO 15189 – Quality & competence requirements for medical laboratories 97
7.1.2.2 CLSI Guidelines (Clinical & Laboratory Standards Institute) – Immunoassay performance & validation 97
7.1.2.3 ISO 13485 – Quality management systems for IVD manufacturers 97
7.1.2.4 ISO 17511 – Metrological traceability of calibrators & control materials 97
7.2 SUSTAINABILITY INITIATIVES 97
7.2.1 REAGENT PACKAGING REDUCTION & RECYCLABLE MATERIALS (INDUSTRY INITIATIVES) 98
7.2.2 ENERGY-EFFICIENT & LOW-WASTE ANALYZER OPERATIONS 98
7.2.3 LABORATORY WASTE MANAGEMENT & GREEN LAB PROGRAMS 98
7.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS 98
7.3.1 CERTIFICATIONS & LABELING STANDARDS 98
7.3.2 ECO-STANDARDS & SUSTAINABILITY CONSIDERATIONS 99
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR 100
8.1 DECISION-MAKING PROCESS 100
8.2 KEY STAKEHOLDERS & BUYING CRITERIA 101
8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS 101
8.2.1.1 Influence of stakeholders on buying process for product types 101
8.2.2 BUYING CRITERIA 102
8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES 103
8.4 UNMET NEEDS IN VARIOUS END-USE SETTINGS 104
8.5 MARKET PROFITABILITY 105
8.5.1 REVENUE POTENTIAL 105
8.5.2 COST DYNAMICS 105
8.5.3 MARGIN OPPORTUNITIES IN KEY APPLICATIONS 105
9 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE 106
9.1 INTRODUCTION 107
9.2 CONSUMABLES 107
9.2.1 CONSUMABLES TO ACCOUNT FOR LARGEST MARKET SHARE
DURING STUDY PERIOD 107
9.3 INSTRUMENTS 109
9.3.1 INCREASING ADVANCES WITH HIGH-THROUGHPUT CAPACITIES AND GROWING AUTOMATION TRENDS TO DRIVE MARKET 109
10 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY 113
10.1 INTRODUCTION 114
10.2 CHEMILUMINESCENCE ENZYME IMMUNOASSAYS 114
10.2.1 HIGH RELIABILITY OF BLOOD TESTS IN CLINICAL DIAGNOSTICS AND PHARMACEUTICAL RESEARCH TO DRIVE MARKET 114
10.3 ELECTROCHEMILUMINESCENCE IMMUNOASSAYS 116
10.3.1 RISING DEMAND FOR HIGH-SENSITIVITY CARDIAC AND ONCOLOGY BIOMARKERS TO DRIVE MARKET 116
10.4 MICROPARTICLE CHEMILUMINESCENCE IMMUNOASSAYS 118
10.4.1 SUPERIOR SENSITIVITY AND EARLY DISEASE DETECTION TO DRIVE MARKET GROWTH 118
11 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE 120
11.1 INTRODUCTION 121
11.2 BLOOD 121
11.2.1 RELIABILITY OF BLOOD TESTS FOR IMMUNOASSAY DIAGNOSTIC PROCEDURES AND HEALTH SCREENING TO DRIVE MARKET 121
11.3 URINE 122
11.3.1 INCREASING USE IN LAW ENFORCEMENT AND DRUG TESTING CENTERS TO DRIVE MARKET 122
11.4 SALIVA 124
11.4.1 CONVENIENCE AND EASY TESTING OF BIOLOGICALLY ACTIVE PARTS OF HORMONES TO DRIVE MARKET 124
11.5 OTHER SAMPLE TYPES 125
12 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION 126
12.1 INTRODUCTION 127
12.2 INFECTIOUS DISEASES 128
12.2.1 RISING PREVALENCE OF INFECTIOUS DISEASES TO DRIVE MARKET 128
12.3 ENDOCRINOLOGY 129
12.3.1 RISING INCIDENCES OF DIABETES TO PROPEL DEMAND FOR DIAGNOSTICS 129
12.4 ONCOLOGY 131
12.4.1 RISING BURDEN OF CANCER AND EMPHASIS ON EARLY
DISEASE DIAGNOSIS TO DRIVE MARKET 131
12.5 CARDIOLOGY 133
12.5.1 HIGH BURDEN OF CARDIOVASCULAR DISEASES AMONG ALL AGE GROUPS TO DRIVE MARKET 133
12.6 ALLERGY DIAGNOSTICS 135
12.6.1 GROWING PREVALENCE OF ALLERGIES TO INCREASE DEMAND FOR CHEMILUMINESCENCE IMMUNOASSAYS 135
12.7 BLOOD SCREENING 136
12.7.1 RISING VOLUME OF BLOOD DONATIONS AND INCREASING INCIDENCES OF BLOOD-RELATED DISORDERS TO DRIVE MARKET 136
12.8 AUTOIMMUNE DISORDERS 138
12.8.1 GROWING PREVALENCE OF AUTOIMMUNE DISORDERS AND RISING ECONOMIC BURDEN OF DISEASE MANAGEMENT TO DRIVE MARKET 138
12.9 BONE & MINERAL DISORDERS 140
12.9.1 HIGH DISORDER PREVALENCE TO OFFER STRONG GROWTH OPPORTUNITIES FOR IMMUNOASSAYS 140
12.10 TOXICOLOGY 142
12.10.1 INCREASED DRUG-OF-ABUSE TESTING AND HIGHER ILLICIT DRUG CONSUMPTION TO DRIVE MARKET 142
12.11 NEWBORN SCREENING 144
12.11.1 ROUTINE USE IN FIRST-TIER NEWBORN SCREENING PROTOCOL TO SUSTAIN DEMAND 144
12.12 THERAPEUTIC DRUG MONITORING 145
12.12.1 RAPID DETECTION TIME AND GOOD SPECIFICITY TO DRIVE MARKET 145
12.13 METABOLIC DISORDERS 147
12.13.1 RISING PREVALENCE OF METABOLIC DISORDERS TO DRIVE MARKET 147
12.14 GASTROENTEROLOGY 149
12.14.1 INCREASING INCIDENCE OF GASTROINTESTINAL TRACT INFECTIONS AND SERIOUS GASTRIC DISEASES TO DRIVE MARKET 149
12.15 NEUROLOGY 150
12.15.1 EFFECTIVE DETECTION, MONITORING, AND MANAGEMENT OF NEUROLOGICAL DISORDERS TO DRIVE SEGMENT 150
12.16 RESPIRATORY DISEASES 152
12.16.1 EASY DETECTION AND DIAGNOSIS OF RESPIRATORY
DISEASES TO DRIVE MARKET 152
12.17 OTHER APPLICATIONS 153
13 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER 155
13.1 INTRODUCTION 156
13.2 HOSPITALS 156
13.2.1 GROWING PATIENT POPULATION AND TESTING VOLUMES ENSURE LEADERSHIP OF HOSPITALS 156
13.3 CLINICAL LABORATORIES 159
13.3.1 RISING CLINICAL TEST VOLUMES AND REQUIREMENTS TO DRIVE SEGMENT 159
13.4 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES AND CROS 160
13.4.1 RISING DRUG DISCOVERY ACTIVITY AND CLINICAL STUDIES
TO DRIVE MARKET 160
13.5 OTHER END USERS 162
14 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY REGION 164
14.1 INTRODUCTION 165
14.1.1 GLOBAL: VOLUME ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS, 2024–2031 (HUNDRED UNITS) 165
14.2 NORTH AMERICA 166
14.2.1 MACROECONOMIC OUTLOOK 166
14.2.2 NORTH AMERICA: VOLUME ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS, 2024–2031 (HUNDRED UNITS) 169
14.2.3 US 172
14.2.3.1 US to hold largest share of North American market during forecast period 172
14.2.4 CANADA 176
14.2.4.1 Rising government initiatives and funding for early disease diagnosis to drive market 176
14.3 EUROPE 181
14.3.1 MACROECONOMIC OUTLOOK 182
14.3.2 EUROPE: VOLUME ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS, 2024–2031 (HUNDRED UNITS) 184
14.3.3 GERMANY 187
14.3.3.1 Growing investments in clinical diagnostics research and increasing government spending on healthcare to drive market 187
14.3.4 FRANCE 192
14.3.4.1 Favorable reimbursement policies and increased investments in diagnostics to drive market 192
14.3.5 UK 197
14.3.5.1 Increasing number of accredited clinical laboratories and growing accessibility to IVD tests to drive market 197
14.3.6 ITALY 202
14.3.6.1 Growing geriatric population and increasing support for research to drive market 202
14.3.7 SPAIN 206
14.3.7.1 Rising adoption of technologically advanced immunoassay systems to drive market 206
14.3.8 REST OF EUROPE 210
14.4 ASIA PACIFIC 214
14.4.1 MACROECONOMIC OUTLOOK 215
14.4.2 ASIA PACIFIC: VOLUME ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS, 2024–2031 (HUNDRED UNITS) 218
14.4.3 JAPAN 221
14.4.3.1 Growing investments in clinical diagnostics research to drive market 221
14.4.4 CHINA 225
14.4.4.1 China to register highest growth rate in Asia Pacific market during study period 225
14.4.5 INDIA 230
14.4.5.1 Growing medical tourism and healthcare infrastructure
to drive market 230
14.4.6 SOUTH KOREA 234
14.4.6.1 Rising healthcare spending for innovative IVD technologies
to drive market 234
14.4.7 AUSTRALIA 238
14.4.7.1 Rising healthcare spending for innovative IVD technologies
to drive market 238
14.4.8 REST OF ASIA PACIFIC 242
14.5 LATIN AMERICA 246
14.5.1 MACROECONOMIC OUTLOOK 247
14.5.2 LATIN AMERICA: VOLUME ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS, 2024–2031 (HUNDRED UNITS) 248
14.5.3 BRAZIL 251
14.5.3.1 Improving healthcare facilities to drive market 251
14.5.4 MEXICO 255
14.5.4.1 Robust healthcare system to drive market growth in Mexico 255
14.5.5 REST OF LATIN AMERICA 259
14.6 MIDDLE EAST & AFRICA 263
14.6.1 MACROECONOMIC OUTLOOK 264
14.6.2 MIDDLE EAST & AFRICA: VOLUME ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS, 2024–2031 (HUNDRED UNITS) 265
14.6.3 GCC COUNTRIES 268
14.6.4 REST OF MIDDLE EAST & AFRICA 272
15 COMPETITIVE LANDSCAPE 277
15.1 OVERVIEW 277
15.2 KEY PLAYER STRATEGIES/RIGHT TO WIN, 2023–2026 277
15.3 REVENUE ANALYSIS, 2021–2025 279
15.4 MARKET SHARE ANALYSIS, 2025 280
15.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025 282
15.5.1 STARS 282
15.5.2 EMERGING LEADERS 283
15.5.3 PERVASIVE PLAYERS 283
15.5.4 PARTICIPANTS 283
15.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025 284
15.5.5.1 Company footprint 284
15.5.5.2 Region footprint 285
15.5.5.3 Product type footprint 286
15.5.5.4 Technology footprint 287
15.5.5.5 Sample type footprint 288
15.5.5.6 Application footprint 289
15.6 COMPANY EVALUATION MATRIX: START-UPS/SMES, 2025 291
15.6.1 PROGRESSIVE COMPANIES 291
15.6.2 RESPONSIVE COMPANIES 291
15.6.3 DYNAMIC COMPANIES 291
15.6.4 STARTING BLOCKS 291
15.6.5 COMPETITIVE BENCHMARKING: START-UPS/SMES, 2025 293
15.6.5.1 Detailed list of key start-ups/SMEs 293
15.6.5.2 Competitive benchmarking of key start-ups/SMEs 294
15.7 COMPANY VALUATION AND FINANCIAL METRICS 295
15.8 PRODUCT COMPARISON 296
15.8.1 F. HOFFMANN-LA ROCHE LTD 297
15.8.2 DANAHER CORPORATION 297
15.8.3 ABBOTT 297
15.8.4 SIEMENS HEALTHINEERS AG 297
15.8.5 DIASORIN S.P.A 297
15.9 COMPETITIVE SCENARIO 297
16 COMPANY PROFILES 303
16.1 KEY PLAYERS 303
16.1.1 F. HOFFMANN-LA ROCHE LTD 303
16.1.1.1 Business overview 303
16.1.1.2 Products offered 304
16.1.1.3 Recent developments 305
16.1.1.3.1 Product approvals 305
16.1.1.3.2 Deals 305
16.1.1.4 MnM view 305
16.1.1.4.1 Key strengths/Right to win 305
16.1.1.4.2 Strategic choices 306
16.1.1.4.3 Weaknesses/Competitive threats 306
16.1.2 ABBOTT 307
16.1.2.1 Business overview 307
16.1.2.2 Products offered 308
16.1.2.3 Recent developments 309
16.1.2.3.1 Deals 309
16.1.2.4 MnM view 309
16.1.2.4.1 Key strengths/Right to win 309
16.1.2.4.2 Strategic choices 310
16.1.2.4.3 Weaknesses/Competitive threats 310
16.1.3 SIEMENS HEALTHINEERS AG 311
16.1.3.1 Business overview 311
16.1.3.2 Products offered 312
16.1.3.3 Recent developments 313
16.1.3.3.1 Deals 313
16.1.3.4 MnM view 314
16.1.3.4.1 Key strengths/Right to win 314
16.1.3.4.2 Strategic choices 314
16.1.3.4.3 Weaknesses/Competitive threats 314
16.1.4 DANAHER CORPORATION 315
16.1.4.1 Business overview 315
16.1.4.2 Products offered 316
16.1.4.3 Recent developments 317
16.1.4.3.1 Deals 317
16.1.4.4 MnM view 317
16.1.4.4.1 Key strengths/Right to win 317
16.1.4.4.2 Strategic choices 317
16.1.4.4.3 Weaknesses/Competitive threats 318
16.1.5 DIASORIN S.P.A. 319
16.1.5.1 Business overview 319
16.1.5.2 Products offered 321
16.1.5.3 Recent developments 321
16.1.5.3.1 Product approvals 321
16.1.5.3.2 Deals 321
16.1.5.4 MnM view 322
16.1.5.4.1 Key strengths/Right to win 322
16.1.5.4.2 Strategic choices 322
16.1.5.4.3 Weaknesses/Competitive threats 322
16.1.6 QUIDELORTHO CORPORATION 323
16.1.6.1 Business overview 323
16.1.6.2 Products offered 324
16.1.6.3 Recent developments 325
16.1.6.3.1 Deals 325
16.1.7 SYSMEX CORPORATION 326
16.1.7.1 Business overview 326
16.1.7.2 Products offered 327
16.1.7.3 Recent developments 328
16.1.7.3.1 Product launches 328
16.1.7.3.2 Deals 329
16.1.8 SHENZHEN NEW INDUSTRIES BIOMEDICAL ENGINEERING CO., LTD. 330
16.1.8.1 Business overview 330
16.1.8.2 Products offered 331
16.1.8.3 Recent developments 332
16.1.8.3.1 Product approvals 332
16.1.9 TOSOH CORPORATION 333
16.1.9.1 Business overview 333
16.1.9.2 Products offered 335
16.1.9.3 Recent developments 336
16.1.9.3.1 Product launches 336
16.1.10 WERFEN 337
16.1.10.1 Business overview 337
16.1.10.2 Products offered 338
16.1.10.3 Recent developments 339
16.1.10.3.1 Deals 339
16.1.11 BIO-RAD LABORATORIES, INC. 340
16.1.11.1 Business overview 340
16.1.11.2 Products offered 341
16.1.11.3 Recent developments 342
16.1.11.3.1 Product launches 342
16.1.12 SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD. 343
16.1.12.1 Business overview 343
16.1.12.2 Products offered 343
16.1.12.3 Recent developments 344
16.1.12.3.1 Deals 344
16.1.13 FUJIREBIO 345
16.1.13.1 Business overview 345
16.1.13.2 Products offered 345
16.1.13.3 Recent developments 346
16.1.13.3.1 Deals 346
16.1.14 EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG 347
16.1.14.1 Business overview 347
16.1.14.2 Products offered 347
16.1.14.3 Recent developments 348
16.1.14.3.1 Product launches & approvals 348
16.1.14.3.2 Deals 348
16.1.15 AGAPPE DIAGNOSTICS LTD. 349
16.1.15.1 Business overview 349
16.1.15.2 Products offered 349
16.1.15.3 Recent developments 350
16.1.15.3.1 Deals 350
16.2 OTHER PLAYERS 351
16.2.1 ZECEN BIOTECH CO., LTD 351
16.2.2 MACCURA BIOTECHNOLOGY CO., LTD. 352
16.2.3 AUTOBIO DIAGNOSTICS 353
16.2.4 ARTRON LABORATORIES INC. 354
16.2.5 HUMAN GESELLSCHAFT FÜR BIOCHEMICA UND DIAGNOSTICA MBH 355
16.2.6 ABNOVA CORPORATION 356
16.2.7 BEIJING HOTGEN BIOTECH CO., LTD. 357
16.2.8 ELABSCIENCE 358
16.2.9 GETEIN BIOTECH, INC. 359
16.2.10 SHANGHAI KEHUA BIO-ENGINEERING CO., LTD. 360
17 RESEARCH METHODOLOGY 361
17.1 RESEARCH DATA 361
17.1.1 SECONDARY DATA 362
17.1.1.1 Key data from secondary sources 363
17.1.1.2 Primary sources 363
17.1.1.3 Key data from primary sources 364
17.1.1.4 Breakdown of primaries 364
17.2 MARKET SIZE ESTIMATION 365
17.3 GROWTH FORECAST 369
17.4 MARKET BREAKDOWN & DATA TRIANGULATION 370
17.5 RESEARCH ASSUMPTIONS 372
17.6 RESEARCH LIMITATIONS 372
17.7 RISK ASSESSMENT 372
18 APPENDIX 373
18.1 DISCUSSION GUIDE 373
18.2 KNOWLEDGESTORE: MARKETSANDMARKETS’ SUBSCRIPTION PORTAL 378
18.3 CUSTOMIZATION OPTIONS 380
18.3.1 GEOGRAPHIC ANALYSIS 380
18.3.2 REGIONAL/COUNTRY-LEVEL MARKET SHARE ANALYSIS 380
18.3.3 COMPANY INFORMATION 380
18.3.4 PRODUCT TYPE ANALYSIS 380
18.3.4.1 By Analyzer (Semi and fully automated system) 380
18.3.4.2 By Technology (Breakdown by subcategories) 380
18.3.5 BY END USER ((BREAKDOWN BY SUBCATEGORIES) 380
18.3.6 BY MODE OF PURCHASE/BUSINESS MODEL 380
18.3.7 COUNTRY-LEVEL VOLUME ANALYSIS 380
18.3.8 BY PRODUCT TYPE MARKET SHARE ANALYSIS (TOP 5 PLAYERS) 380
18.3.9 ANY CONSULTS/CUSTOM REQUIREMENTS AS PER CLIENT REQUEST 380
18.4 RELATED REPORTS 381
18.5 AUTHOR DETAILS 382
LIST OF TABLES
TABLE 1 STANDARD CURRENCY CONVERSION RATES (UNIT OF USD) 33
TABLE 2 UNMET CUSTOMER NEEDS & WHITE SPACES IN CHEMILUMINESCENCE IMMUNOASSAY MARKET 56
TABLE 3 INTERCONNECTED MARKETS IN CHEMILUMINESCENCE IMMUNOASSAY MARKET 57
TABLE 4 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS IN CHEMILUMINESCENCE IMMUNOASSAY MARKET 59
TABLE 5 CHEMILUMINESCENCE IMMUNOASSAY MARKET:
PORTER’S FIVE FORCES ANALYSIS 60
TABLE 6 GLOBAL GROSS DOMESTIC PRODUCT AT CURRENT PRICES,
2021–2030 (USD BILLION) 63
TABLE 7 CHEMILUMINESCENCE IMMUNOASSAY MARKET: ROLE IN ECOSYSTEM 69
TABLE 8 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY KEY PLAYER, 2023–2025 (USD) 71
TABLE 9 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY REGION, 2023–2025 (USD) 72
TABLE 10 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY TECHNOLOGY, 2023–2025 (USD) 73
TABLE 11 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY CONSUMABLES (KITS, REAGENTS, AND OTHERS), BY SAMPLE TYPE,
2023–2025 (USD) 73
TABLE 12 IMPORT DATA FOR HS CODE 9027.89-COMPLIANT PRODUCTS, BY COUNTRY, 2021–2025 (USD THOUSAND) 74
TABLE 13 EXPORT DATA FOR HS CODE 901839-COMPLIANT PRODUCTS, BY COUNTRY, 2021–2025 (USD THOUSAND) 75
TABLE 14 DETAILED LIST OF CONFERENCES & EVENTS, 2026–2027 75
TABLE 15 US-ADJUSTED RECIPROCAL TARIFF RATES 80
TABLE 16 CHEMILUMINESCENCE IMMUNOASSAY MARKET:
TECHNOLOGY/PRODUCT ROADMAP 86
TABLE 17 CHEMILUMINESCENCE IMMUNOASSAY MARKET: LIST OF MAJOR PATENTS, JANUARY 2022–SEPTEMBER 2025 88
TABLE 18 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES,
AND OTHER ORGANIZATIONS 94
TABLE 19 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES,
AND OTHER ORGANIZATIONS 95
TABLE 20 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES,
AND OTHER ORGANIZATIONS 95
TABLE 21 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES,
AND OTHER ORGANIZATIONS 96
TABLE 22 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES,
AND OTHER ORGANIZATIONS 96
TABLE 23 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR PRODUCT TYPES 101
TABLE 24 KEY BUYING CRITERIA FOR TOP THREE END USERS 102
TABLE 25 CHEMILUMINESCENCE IMMUNOASSAY MARKET: UNMET NEEDS IN
KEY END-USE SETTINGS 104
TABLE 26 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE,
2024–2031 (USD MILLION) 107
TABLE 27 LIST OF CHEMILUMINESCENCE IMMUNOASSAY CONSUMABLES AVAILABLE 108
TABLE 28 CHEMILUMINESCENCE IMMUNOASSAY CONSUMABLES MARKET, BY COUNTRY, 2024–2031 (USD MILLION) 109
TABLE 29 CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET,
2024–2031 (HUNDRED UNITS) 110
TABLE 30 LIST OF CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS AVAILABLE 111
TABLE 31 CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET, BY COUNTRY, 2024–2031 (USD MILLION) 112
TABLE 32 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 114
TABLE 33 CHEMILUMINESCENCE ENZYME IMMUNOASSAY MARKET, BY COUNTRY,
2024–2031 (USD MILLION) 115
TABLE 34 ELECTROCHEMILUMINESCENCE IMMUNOASSAY MARKET, BY COUNTRY,
2024–2031 (USD MILLION) 117
TABLE 35 MICROPARTICLE CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY COUNTRY, 2024–2031 (USD MILLION) 119
TABLE 36 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 121
TABLE 37 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR BLOOD SAMPLES,
BY COUNTRY, 2024–2031 (USD MILLION) 122
TABLE 38 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR URINE SAMPLES,
BY COUNTRY, 2024–2031 (USD MILLION) 123
TABLE 39 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR SALIVA SAMPLES,
BY REGION, 2024–2031 (USD MILLION) 124
TABLE 40 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR OTHER SAMPLE TYPES,
BY COUNTRY, 2024–2031 (USD MILLION) 125
TABLE 41 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 127
TABLE 42 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR INFECTIOUS DISEASES,
BY COUNTRY, 2024–2031 (USD MILLION) 129
TABLE 43 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR ENDOCRINOLOGY,
BY COUNTRY, 2024–2031 (USD MILLION) 130
TABLE 44 CANCER INCIDENCE, BY TYPE (2022) 131
TABLE 45 INCREASING INCIDENCE OF CANCER, BY REGION, 2020 VS.
2030 VS. 2040 (MILLION) 132
TABLE 46 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR ONCOLOGY,
BY COUNTRY, 2024–2031 (USD MILLION) 133
TABLE 47 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR CARDIOLOGY,
BY COUNTRY, 2024–2031 (USD MILLION) 134
TABLE 48 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR ALLERGY DIAGNOSTICS,
BY COUNTRY, 2024–2031 (USD MILLION) 136
TABLE 49 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR BLOOD SCREENING,
BY COUNTRY, 2024–2031 (USD MILLION) 137
TABLE 50 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR AUTOIMMUNE DISORDERS, BY COUNTRY, 2024–2031 (USD MILLION) 139
TABLE 51 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR BONE & MINERAL DISORDERS, BY COUNTRY, 2024–2031 (USD MILLION) 141
TABLE 52 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR TOXICOLOGY, BY COUNTRY, 2024–2031 (USD MILLION) 143
TABLE 53 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR NEWBORN SCREENING,
BY COUNTRY, 2024–2031 (USD MILLION) 145
TABLE 54 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR THERAPEUTIC DRUG MONITORING, BY COUNTRY, 2024–2031 (USD MILLION) 146
TABLE 55 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR METABOLIC DISORDERS,
BY COUNTRY, 2024–2031 (USD MILLION) 148
TABLE 56 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR GASTROENTEROLOGY,
BY COUNTRY, 2024–2031 (USD MILLION) 150
TABLE 57 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR NEUROLOGY, BY COUNTRY, 2024–2031 (USD MILLION) 151
TABLE 58 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR RESPIRATORY DISEASES,
BY COUNTRY, 2024–2031 (USD MILLION) 153
TABLE 59 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR OTHER APPLICATIONS,
BY COUNTRY, 2024–2031 (USD MILLION) 154
TABLE 60 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 156
TABLE 61 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR HOSPITALS,
BY COUNTRY, 2024–2031 (USD MILLION) 158
TABLE 62 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR CLINICAL LABORATORIES
BY COUNTRY, 2024–2031 (USD MILLION) 160
TABLE 63 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES AND CROS, BY COUNTRY,
2024–2031 (USD MILLION) 161
TABLE 64 CHEMILUMINESCENCE IMMUNOASSAY MARKET FOR OTHER END USERS,
BY COUNTRY, 2024–2031 (USD MILLION) 163
TABLE 65 CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY REGION,
2024–2031 (USD MILLION) 165
TABLE 66 CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET,
2024–2031 (HUNDRED UNITS) 165
TABLE 67 NORTH AMERICA: MACROECONOMIC OUTLOOK 167
TABLE 68 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY COUNTRY, 2024–2031 (USD MILLION) 168
TABLE 69 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 169
TABLE 70 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET, 2024–2031 (HUNDRED UNITS) 169
TABLE 71 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 170
TABLE 72 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY SAMPLE TYPE, 2024–2031 (USD MILLION) 170
TABLE 73 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 171
TABLE 74 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY END USER, 2024–2031 (USD MILLION) 172
TABLE 75 US: KEY MACROINDICATORS 173
TABLE 76 US: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE,
2024–2031 (USD MILLION) 173
TABLE 77 US: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 174
TABLE 78 US: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 174
TABLE 79 US: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 175
TABLE 80 US: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 176
TABLE 81 CANADA: ESTIMATED PREVALENCE OF DIABETES 176
TABLE 82 CANADA: KEY MACROINDICATORS (2024) 177
TABLE 83 CANADA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 178
TABLE 84 CANADA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 178
TABLE 85 CANADA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 179
TABLE 86 CANADA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 180
TABLE 87 CANADA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 181
TABLE 88 EUROPE: MACROECONOMIC OUTLOOK 182
TABLE 89 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY COUNTRY,
2024–2031 (USD MILLION) 183
TABLE 90 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 183
TABLE 91 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET,
2024–2031 (HUNDRED UNITS) 184
TABLE 92 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 184
TABLE 93 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 185
TABLE 94 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 186
TABLE 95 EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 187
TABLE 96 GERMANY: KEY MACROINDICATORS 188
TABLE 97 GERMANY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 189
TABLE 98 GERMANY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 189
TABLE 99 GERMANY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 190
TABLE 100 GERMANY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 191
TABLE 101 GERMANY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 192
TABLE 102 FRANCE: KEY MACROINDICATORS (2024) 193
TABLE 103 FRANCE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 194
TABLE 104 FRANCE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 194
TABLE 105 FRANCE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 195
TABLE 106 FRANCE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 196
TABLE 107 FRANCE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 197
TABLE 108 UK: KEY MACROINDICATORS 198
TABLE 109 UK: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE,
2024–2031 (USD MILLION) 199
TABLE 110 UK: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 199
TABLE 111 UK: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 200
TABLE 112 UK: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 201
TABLE 113 UK: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 202
TABLE 114 ITALY: KEY MACROINDICATORS (2024) 203
TABLE 115 ITALY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE,
2024–2031 (USD MILLION) 203
TABLE 116 ITALY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 204
TABLE 117 ITALY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 204
TABLE 118 ITALY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 205
TABLE 119 ITALY: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 206
TABLE 120 SPAIN: KEY MACROINDICATORS (2024) 207
TABLE 121 SPAIN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 207
TABLE 122 SPAIN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 208
TABLE 123 SPAIN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 208
TABLE 124 SPAIN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 209
TABLE 125 SPAIN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 210
TABLE 126 REST OF EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY PRODUCT TYPE, 2024–2031 (USD MILLION) 211
TABLE 127 REST OF EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 211
TABLE 128 REST OF EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 212
TABLE 129 REST OF EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 213
TABLE 130 REST OF EUROPE: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY END USER, 2024–2031 (USD MILLION) 214
TABLE 131 ASIA PACIFIC: MACROECONOMIC OUTLOOK 215
TABLE 132 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY COUNTRY, 2024–2031 (USD MILLION) 217
TABLE 133 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 217
TABLE 134 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET, 2024–2031 (HUNDRED UNITS) 218
TABLE 135 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 218
TABLE 136 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 219
TABLE 137 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 220
TABLE 138 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER, 2024–2031 (USD MILLION) 221
TABLE 139 JAPAN: KEY MACROINDICATORS (2024) 222
TABLE 140 JAPAN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 222
TABLE 141 JAPAN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 223
TABLE 142 JAPAN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 223
TABLE 143 JAPAN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 224
TABLE 144 JAPAN: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 225
TABLE 145 CHINA: KEY MACROINDICATORS (2024) 227
TABLE 146 CHINA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 227
TABLE 147 CHINA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 228
TABLE 148 CHINA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 228
TABLE 149 CHINA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 229
TABLE 150 CHINA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 230
TABLE 151 INDIA: KEY MACROINDICATORS (2024) 231
TABLE 152 INDIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE,
2024–2031 (USD MILLION) 231
TABLE 153 INDIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 232
TABLE 154 INDIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 232
TABLE 155 INDIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 233
TABLE 156 INDIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 234
TABLE 157 SOUTH KOREA: KEY MACROINDICATORS (2024) 235
TABLE 158 SOUTH KOREA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 235
TABLE 159 SOUTH KOREA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 236
TABLE 160 SOUTH KOREA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 236
TABLE 161 SOUTH KOREA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 237
TABLE 162 SOUTH KOREA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER, 2024–2031 (USD MILLION) 238
TABLE 163 AUSTRALIA: KEY MACROINDICATORS (2024) 239
TABLE 164 AUSTRALIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 239
TABLE 165 AUSTRALIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 240
TABLE 166 AUSTRALIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 240
TABLE 167 AUSTRALIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 241
TABLE 168 AUSTRALIA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER, 2024–2031 (USD MILLION) 242
TABLE 169 REST OF ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY PRODUCT TYPE, 2024–2031 (USD MILLION) 243
TABLE 170 REST OF ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 243
TABLE 171 REST OF ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY SAMPLE TYPE, 2024–2031 (USD MILLION) 244
TABLE 172 REST OF ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 245
TABLE 173 REST OF ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY END USER, 2024–2031 (USD MILLION) 246
TABLE 174 LATIN AMERICA: MACROECONOMIC OUTLOOK 247
TABLE 175 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY COUNTRY, 2024–2031 (USD MILLION) 247
TABLE 176 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 248
TABLE 177 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET, 2024–2031 (HUNDRED UNITS) 248
TABLE 178 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 249
TABLE 179 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY SAMPLE TYPE, 2024–2031 (USD MILLION) 249
TABLE 180 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 250
TABLE 181 LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY END USER, 2024–2031 (USD MILLION) 251
TABLE 182 BRAZIL: KEY MACROINDICATORS (2024) 252
TABLE 183 BRAZIL: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 252
TABLE 184 BRAZIL: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY,
2024–2031 (USD MILLION) 253
TABLE 185 BRAZIL: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE,
2024–2031 (USD MILLION) 253
TABLE 186 BRAZIL: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 254
TABLE 187 BRAZIL: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 255
TABLE 188 MEXICO: KEY MACROINDICATORS (2024) 256
TABLE 189 MEXICO: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 256
TABLE 190 MEXICO: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 257
TABLE 191 MEXICO: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 257
TABLE 192 MEXICO: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION,
2024–2031 (USD MILLION) 258
TABLE 193 MEXICO: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER,
2024–2031 (USD MILLION) 259
TABLE 194 REST OF LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY PRODUCT TYPE, 2024–2031 (USD MILLION) 260
TABLE 195 REST OF LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 260
TABLE 196 REST OF LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY SAMPLE TYPE, 2024–2031 (USD MILLION) 261
TABLE 197 REST OF LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 262
TABLE 198 REST OF LATIN AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY END USER, 2024–2031 (USD MILLION) 263
TABLE 199 MIDDLE EAST & AFRICA: MACROECONOMIC OUTLOOK 264
TABLE 200 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY REGION, 2024–2031 (USD MILLION) 264
TABLE 201 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY PRODUCT TYPE, 2024–2031 (USD MILLION) 265
TABLE 202 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY INSTRUMENTS MARKET, 2024–2031 (HUNDRED UNITS) 265
TABLE 203 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 266
TABLE 204 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY SAMPLE TYPE, 2024–2031 (USD MILLION) 266
TABLE 205 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 267
TABLE 206 MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY END USER, 2024–2031 (USD MILLION) 268
TABLE 207 GCC COUNTRIES: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 269
TABLE 208 GCC COUNTRIES: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY TECHNOLOGY, 2024–2031 (USD MILLION) 269
TABLE 209 GCC COUNTRIES: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY SAMPLE TYPE, 2024–2031 (USD MILLION) 270
TABLE 210 GCC COUNTRIES: CHEMILUMINESCENCE IMMUNOASSAY MARKET,
BY APPLICATION, 2024–2031 (USD MILLION) 271
TABLE 211 GCC COUNTRIES: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER, 2024–2031 (USD MILLION) 272
TABLE 212 REST OF MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY PRODUCT TYPE, 2024–2031 (USD MILLION) 273
TABLE 213 REST OF MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY TECHNOLOGY, 2024–2031 (USD MILLION) 273
TABLE 214 REST OF MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY SAMPLE TYPE, 2024–2031 (USD MILLION) 274
TABLE 215 REST OF MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY APPLICATION, 2024–2031 (USD MILLION) 275
TABLE 216 REST OF MIDDLE EAST & AFRICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET, BY END USER, 2024–2031 (USD MILLION) 276
TABLE 217 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN CHEMILUMINESCENCE IMMUNOASSAY MARKET, JANUARY 2023–JANUARY 2026 278
TABLE 218 CHEMILUMINESCENCE IMMUNOASSAY MARKET: DEGREE OF COMPETITION, 2025 281
TABLE 219 CHEMILUMINESCENCE IMMUNOASSAY MARKET: REGION FOOTPRINT 285
TABLE 220 CHEMILUMINESCENCE IMMUNOASSAY MARKET: PRODUCT TYPE FOOTPRINT 286
TABLE 221 CHEMILUMINESCENCE IMMUNOASSAY MARKET: TECHNOLOGY FOOTPRINT 287
TABLE 222 CHEMILUMINESCENCE IMMUNOASSAY MARKET: SAMPLE TYPE FOOTPRINT 288
TABLE 223 CHEMILUMINESCENCE IMMUNOASSAY MARKET: APPLICATION FOOTPRINT 289
TABLE 224 CHEMILUMINESCENCE IMMUNOASSAY MARKET: DETAILED LIST
OF KEY START-UPS/SMES 293
TABLE 225 CHEMILUMINESCENCE IMMUNOASSAY MARKET: COMPETITIVE
BENCHMARKING OF KEY START-UPS/SMES 294
TABLE 226 CHEMILUMINESCENCE IMMUNOASSAY MARKET: PRODUCT
LAUNCHES & APPROVALS, JANUARY 2023–JANUARY 2026 298
TABLE 227 CHEMILUMINESCENCE IMMUNOASSAY MARKET: DEALS,
JANUARY 2023–JANUARY 2026 300
TABLE 228 F. HOFFMANN-LA ROCHE LTD: COMPANY OVERVIEW 303
TABLE 229 F. HOFFMANN-LA ROCHE LTD: PRODUCTS OFFERED 304
TABLE 230 F. HOFFMANN-LA ROCHE LTD: PRODUCT APPROVALS,
JANUARY 2023–JANUARY 2026 305
TABLE 231 F. HOFFMANN-LA ROCHE LTD: DEALS, JANUARY 2023–JANUARY 2026 305
TABLE 232 ABBOTT: COMPANY OVERVIEW 307
TABLE 233 ABBOTT: PRODUCTS OFFERED 308
TABLE 234 ABBOTT: DEALS, JANUARY 2023–JANUARY 2026 309
TABLE 235 SIEMENS HEALTHINEERS AG: COMPANY OVERVIEW 311
TABLE 236 SIEMENS HEALTHINEERS AG: PRODUCTS OFFERED 312
TABLE 237 SIEMENS HEALTHINEERS AG: DEALS, JANUARY 2023–JANUARY 2026 313
TABLE 238 DANAHER CORPORATION: COMPANY OVERVIEW 315
TABLE 239 DANAHER CORPORATION: PRODUCTS OFFERED 316
TABLE 240 DANAHER CORPORATION: DEALS, JANUARY 2023–JANUARY 2026 317
TABLE 241 DIASORIN S.P.A.: COMPANY OVERVIEW 319
TABLE 242 DIASORIN S.P.A.: PRODUCTS OFFERED 321
TABLE 243 DIASORIN S.P.A.: PRODUCT APPROVALS, JANUARY 2023–JANUARY 2026 321
TABLE 244 DIASORIN S.P.A.: DEALS, JANUARY 2023–JANUARY 2026 321
TABLE 245 QUIDELORTHO CORPORATION: COMPANY OVERVIEW 323
TABLE 246 QUIDELORTHO CORPORATION: PRODUCTS OFFERED 324
TABLE 247 QUIDELORTHO CORPORATION: DEALS, JANUARY 2023–JANUARY 2026 325
TABLE 248 SYSMEX CORPORATION: COMPANY OVERVIEW 326
TABLE 249 SYSMEX CORPORATION: PRODUCTS OFFERED 327
TABLE 250 SYSMEX CORPORATION: PRODUCT LAUNCHES, JANUARY 2023–JANUARY 2026 328
TABLE 251 SYSMEX CORPORATION: DEALS, JANUARY 2023–JANUARY 2026 329
TABLE 252 SHENZHEN NEW INDUSTRIES BIOMEDICAL ENGINEERING CO., LTD.:
COMPANY OVERVIEW 330
TABLE 253 SHENZHEN NEW INDUSTRIES BIOMEDICAL ENGINEERING CO., LTD.:
PRODUCTS OFFERED 331
TABLE 254 SHENZHEN NEW INDUSTRIES BIOMEDICAL ENGINEERING CO., LTD.:
PRODUCT APPROVALS, JANUARY 2023–JANUARY 2026 332
TABLE 255 TOSOH CORPORATION: COMPANY OVERVIEW 333
TABLE 256 TOSOH CORPORATION: PRODUCTS OFFERED 335
TABLE 257 TOSOH CORPORATION: PRODUCT LAUNCHES, JANUARY 2023–JANUARY 2026 336
TABLE 258 WERFEN: COMPANY OVERVIEW 337
TABLE 259 WERFEN: PRODUCTS OFFERED 338
TABLE 260 WERFEN: DEALS, JANUARY 2023–JANUARY 2026 339
TABLE 261 BIO-RAD LABORATORIES, INC.: COMPANY OVERVIEW 340
TABLE 262 BIO-RAD LABORATORIES, INC.: PRODUCTS OFFERED 341
TABLE 263 BIO-RAD LABORATORIES, INC.: PRODUCT LAUNCHES,
JANUARY 2023–JANUARY 2026 342
TABLE 264 SHENZHEN MINDRAY BIOMEDICAL ELECTRONICS CO., LTD.: COMPANY OVERVIEW 343
TABLE 265 SHENZHEN MINDRAY BIOMEDICAL ELECTRONICS CO., LTD.: PRODUCTS OFFERED 343
TABLE 266 SHENZHEN MINDRAY BIOMEDICAL ELECTRONICS CO., LTD.: DEALS, JANUARY 2023–JANUARY 2026 344
TABLE 267 FUJIREBIO: COMPANY OVERVIEW 345
TABLE 268 FUJIREBIO: PRODUCTS OFFERED 345
TABLE 269 FUJIREBIO: DEALS, JANUARY 2023–JANUARY 2026 346
TABLE 270 EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG: COMPANY OVERVIEW 347
TABLE 271 EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG: PRODUCTS OFFERED 347
TABLE 272 EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG: PRODUCT LAUNCHES & APPROVALS, JANUARY 2023–JANUARY 2026 348
TABLE 273 EUROIMMUN MEDIZINISCHE LABORDIAGNOSTIKA AG: DEALS,
JANUARY 2023–JANUARY 2026 348
TABLE 274 AGAPPE DIAGNOSTICS LTD.: COMPANY OVERVIEW 349
TABLE 275 AGAPPE DIAGNOSTICS LTD.: PRODUCTS OFFERED 349
TABLE 276 AGAPPE DIAGNOSTICS LTD.: DEALS, JANUARY 2023–JANUARY 2026 350
TABLE 277 ZECEN BIOTECH CO., LTD: COMPANY OVERVIEW 351
TABLE 278 MACCURA BIOTECHNOLOGY CO., LTD.: COMPANY OVERVIEW 352
TABLE 279 AUTOBIO DIAGNOSTICS: COMPANY OVERVIEW 353
TABLE 280 ARTRON LABORATORIES INC.: COMPANY OVERVIEW 354
TABLE 281 HUMAN GESELLSCHAFT FÜR BIOCHEMICA UND DIAGNOSTICA MBH:
COMPANY OVERVIEW 355
TABLE 282 ABNOVA CORPORATION: COMPANY OVERVIEW 356
TABLE 283 BEIJING HOTGEN BIOTECH CO., LTD.: COMPANY OVERVIEW 357
TABLE 284 ELABSCIENCE: COMPANY OVERVIEW 358
TABLE 285 GETEIN BIOTECH, INC.: COMPANY OVERVIEW 359
TABLE 286 SHANGHAI KEHUA BIO-ENGINEERING CO., LTD.: COMPANY OVERVIEW 360
LIST OF FIGURES
FIGURE 1 CHEMILUMINESCENCE IMMUNOASSAY MARKET SEGMENTATION &
REGIONAL SCOPE 31
FIGURE 2 MARKET SCENARIO 35
FIGURE 3 GLOBAL CHEMILUMINESCENCE IMMUNOASSAY MARKET, 2024–2031 36
FIGURE 4 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN CHEMILUMINESCENCE IMMUNOASSAY MARKET, 2023–2026 36
FIGURE 5 DISRUPTIONS INFLUENCING GROWTH OF CHEMILUMINESCENCE
IMMUNOASSAY (CLIA) MARKET 37
FIGURE 6 HIGH-GROWTH SEGMENTS IN CHEMILUMINESCENCE IMMUNOASSAY MARKET, 2026–2031 38
FIGURE 7 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN THE CHEMILUMINESCENCE IMMUNOASSAY MARKET, IN TERMS OF VALUE, DURING FORECAST PERIOD 39
FIGURE 8 TECHNOLOGICAL ADVANCEMENTS IN MEDICAL DEVICE MODALITIES
TO DRIVE THE MARKET 41
FIGURE 9 CHINA AND CONSUMABLES TO DOMINATE APAC MARKET IN 2025 42
FIGURE 10 CHINA TO REGISTER HIGHEST GROWTH RATE DURING FORECAST PERIOD 43
FIGURE 11 ASIA PACIFIC TO REGISTER HIGHEST GROWTH RATE DURING FORECAST PERIOD 44
FIGURE 12 NORTH AMERICA TO DOMINATE MARKET IN 2030 45
FIGURE 13 CHEMILUMINESCENCE IMMUNOASSAY MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES 47
FIGURE 14 GLOBAL DIABETES INCIDENCE, 2021 VS. 2045 (MILLION) 48
FIGURE 15 NUMBER OF PEOPLE WITH CHRONIC CONDITIONS IN US, 1995–2030 (MILLION INDIVIDUALS) 49
FIGURE 16 GLOBAL GERIATRIC POPULATION, BY REGION, 2019 VS. 2050 (MILLION) 51
FIGURE 17 CHEMILUMINESCENCE IMMUNOASSAY MARKET:
PORTER’S FIVE FORCES ANALYSIS 61
FIGURE 18 CHEMILUMINESCENCE IMMUNOASSAY MARKET: VALUE CHAIN ANALYSIS (2025) 66
FIGURE 19 CHEMILUMINESCENCE IMMUNOASSAY MARKET: SUPPLY CHAIN ANALYSIS (2025) 68
FIGURE 20 CHEMILUMINESCENCE IMMUNOASSAY MARKET: ECOSYSTEM ANALYSIS, 2025 69
FIGURE 21 AVERAGE SELLING PRICE TREND OF CHEMILUMINESCENCE IMMUNOASSAY ANALYZERS, BY REGION, 2023–2025 (USD) 72
FIGURE 22 TRENDS & DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES 76
FIGURE 23 INVESTMENT/VENTURE CAPITAL SCENARIO IN CHEMILUMINESCENCE IMMUNOASSAY MARKET, 2022–2025 77
FIGURE 24 PATENT PUBLICATION TRENDS, JURISDICTION, AND TOP APPLICANT ANALYSIS (JANUARY 2015–DECEMBER 2025) 87
FIGURE 25 TOP APPLICANT COUNTRIES/REGIONS FOR CHEMILUMINESCENCE IMMUNOASSAY PATENTS (JANUARY 2015–DECEMBER 2025) 88
FIGURE 26 MARKET POTENTIAL OF AI/GEN AI ON CLIA ACROSS END USERS 90
FIGURE 27 IMPACT OF AI/GEN AI ON INTERCONNECTED & ADJACENT ECOSYSTEMS 92
FIGURE 28 CHEMILUMINESCENCE IMMUNOASSAY MARKET: DECISION-MAKING FACTORS 101
FIGURE 29 KEY BUYING CRITERIA FOR TOP THREE END USERS 102
FIGURE 30 ADOPTION BARRIERS & INTERNAL CHALLENGES 103
FIGURE 31 GLOBAL INCIDENCE OF DIABETES 147
FIGURE 32 NORTH AMERICA: CHEMILUMINESCENCE IMMUNOASSAY MARKET SNAPSHOT 168
FIGURE 33 ASIA PACIFIC: CHEMILUMINESCENCE IMMUNOASSAY MARKET SNAPSHOT 216
FIGURE 34 CHEMILUMINESCENCE IMMUNOASSAY MARKET: REVENUE ANALYSIS OF TOP FIVE PLAYERS, 2021–2025 279
FIGURE 35 MARKET SHARE ANALYSIS OF COMPANIES OFFERING CHEMILUMINESCENCE IMMUNOASSAYS, 2025 280
FIGURE 36 CHEMILUMINESCENCE IMMUNOASSAY MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025 283
FIGURE 37 CHEMILUMINESCENCE IMMUNOASSAY MARKET: COMPANY FOOTPRINT 284
FIGURE 38 CHEMILUMINESCENCE IMMUNOASSAY MARKET: COMPANY EVALUATION MATRIX (START-UPS/SMES), 2025 292
FIGURE 39 5-YEAR STOCK BETA OF KEY VENDORS 295
FIGURE 40 FINANCIAL METRICS (EV/EBITDA) 295
FIGURE 41 CHEMILUMINESCENCE IMMUNOASSAYS: PRODUCT COMPARATIVE ANALYSIS 296
FIGURE 42 F. HOFFMANN-LA ROCHE LTD: COMPANY SNAPSHOT (2025) 304
FIGURE 43 ABBOTT: COMPANY SNAPSHOT (2025) 308
FIGURE 44 SIEMENS HEALTHINEERS AG: COMPANY SNAPSHOT (2025) 312
FIGURE 45 DANAHER CORPORATION: COMPANY SNAPSHOT (2025) 316
FIGURE 46 DIASORIN S.P.A.: COMPANY SNAPSHOT (2025) 320
FIGURE 47 QUIDELORTHO CORPORATION: COMPANY SNAPSHOT (2025) 324
FIGURE 48 SYSMEX CORPORATION: COMPANY SNAPSHOT (2025) 327
FIGURE 49 SHENZHEN NEW INDUSTRIES BIOMEDICAL ENGINEERING CO., LTD.:
COMPANY SNAPSHOT (2024) 331
FIGURE 50 TOSOH CORPORATION: COMPANY SNAPSHOT (2025) 334
FIGURE 51 WERFEN: COMPANY SNAPSHOT (2024) 338
FIGURE 52 BIO-RAD LABORATORIES, INC.: COMPANY SNAPSHOT (2025) 341
FIGURE 53 RESEARCH DESIGN 361
FIGURE 54 BREAKDOWN OF PRIMARY INTERVIEWS (SUPPLY SIDE): BY COMPANY TYPE, DESIGNATION, AND REGION 364
FIGURE 55 BREAKDOWN OF PRIMARY INTERVIEWS (DEMAND SIDE): BY END USER, DESIGNATION, AND REGION 365
FIGURE 56 MARKET SIZE ESTIMATION PROCESS 366
FIGURE 57 MARKET SIZE ESTIMATION: REVENUE SHARE ANALYSIS (2025) 367
FIGURE 58 CHEMILUMINESCENCE IMMUNOASSAY MARKET: SUPPLY-SIDE ANALYSIS 367
FIGURE 59 TOP-DOWN APPROACH 368
FIGURE 60 CAGR PROJECTIONS FROM ANALYSIS OF DRIVERS, RESTRAINTS,
OPPORTUNITIES, AND CHALLENGES IN CHEMILUMINESCENCE IMMUNOASSAY MARKET (2026–2031) 369
FIGURE 61 CAGR PROJECTIONS: SUPPLY-SIDE ANALYSIS OF CHEMILUMINESCENCE IMMUNOASSAY MARKET (2025) 370
FIGURE 62 DATA TRIANGULATION 371