Nuclear Medicine Market - Global Forecast to 2030
核医学市場 - タイプ[SPECT(Tc-99m、I-123、Ga-67)、PET(F-18)、アルファ線源、ベータ線源(Y-90、Lu-177)、密封小線源療法]、用途(腫瘍学、心臓学、神経学)、手順、エンドユーザー(イメージングセンター、病院)、および地域別 - 2030年までの世界予測
Nuclear Medicine Market by Type [SPECT (Tc-99m, I-123, Ga-67), PET (F-18), Alpha Emitters, Beta Emitters (Y-90, Lu-177), Brachytherapy], Application (Oncology, Cardiology, Neurology), Procedure, End User (Imaging Center, Hospital), and Region – Global Forecast to 2030
| 出版 | MarketsandMarkets |
| 出版年月 | 2026年01月 |
| ページ数 | 536 |
| 図表数 | 589 |
| 価格 | 記載以外のライセンスについてはお問合せください |
| シングルユーザ | USD 4,950 |
| 種別 | 英文調査報告書 |
| 商品番号 | SMR-1461514615 |
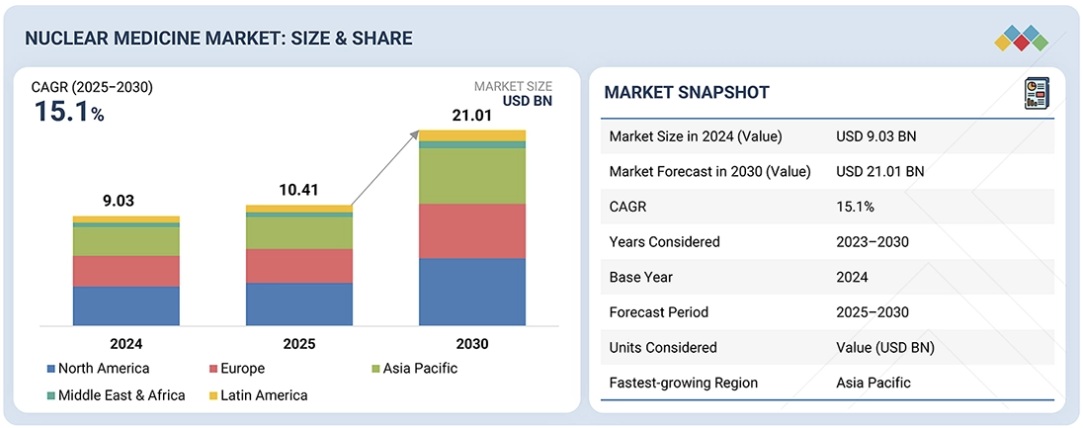
世界の核医学市場は、2025年の104億1,000万米ドルから2030年には210億1,000万米ドルに達すると予測されており、予測期間中は年平均成長率(CAGR)15.1%で成長します。この予測は、腫瘍学、心臓病学、神経学の分野における診断用および治療用放射性医薬品と医療用放射性同位元素の需要増加に牽引されています。さらに、標的放射性核種療法の利用増加、サイクロトロンおよび原子炉ベースの放射性同位元素製造の増加、そして核薬局ネットワークを通じたこれらの製品の入手性向上が、市場の成長を促進しています。さらに、新しい放射性医薬品が絶えず開発され、規制当局によって承認されているため、世界中で放射性医薬品の使用が大幅に増加しています。こうした放射性医薬品の使用増加が、市場の成長をさらに促進しています。
調査範囲
本レポートは、核医学市場を分析し、様々な市場セグメントの市場規模と将来の成長可能性を、種類、用途、数量評価、エンドユーザー、地域別に推定することを目的としています。また、この市場で活動する主要企業の競合分析に加え、各社の企業概要、製品ラインナップ、最近の動向、主要な市場戦略についても解説しています。
レポートを購入する理由
このレポートは、既存企業だけでなく新規参入企業や中小企業にとっても、市場の動向を把握する上で有益な情報を提供し、ひいては市場シェアの拡大に貢献します。レポートを購入した企業は、以下の戦略のいずれか、または組み合わせて活用することで、市場における自社の地位を強化することができます。
本レポートは、以下の点について洞察を提供します。
- 核医学市場の成長に影響を与える主要な推進要因(標的疾患の発生率および有病率の増加、アルファ線放射免疫療法に基づく標的癌治療の開発、Mo-99の需給ギャップ縮小に向けた取り組み)、制約要因(放射性医薬品の短い半減期)、機会(神経学的用途における放射性医薬品の使用、新興国における成長機会)、課題(病院予算の削減と高額な設備費)の分析
- 製品開発/イノベーション:核医学市場における今後の技術、研究開発活動、および製品発売に関する詳細な洞察
- 市場開発:地域全体にわたる収益性の高い新興市場(タイプ、用途、数量評価、およびエンドユーザーに基づく)に関する包括的な情報
- 市場の多様化:核医学市場における製品ポートフォリオ、成長地域、最近の開発状況、および投資に関する網羅的な情報
- 競合評価:市場シェア、成長戦略、製品核医学市場で活動する主要企業[GEヘルスケア(米国)、カーディナルヘルス(米国)、キュリウム(フランス)、バイエルAG(ドイツ)、ランテウスホールディングス(米国)]の提供内容と能力
Report Description
The global nuclear medicine market is projected to reach USD 21.01 billion by 2030, growing from USD 10.41 billion in 2025 at a CAGR of 15.1% during the forecast period. This projection is driven by the growing demand for diagnostic and therapeutic radiopharmaceuticals and medical radioisotopes in the oncology, cardiology, and neurology fields. Additionally, the increased utilization of targeted radionuclide therapies, growth in cyclotron and reactor-based production of radioisotopes, and enhanced availability of these products through nuclear pharmacy networks are enhancing the growth of the market. Moreover, there is a significant increase in the use of radiopharmaceuticals across the globe as new ones are constantly developed and approved by regulatory bodies. This increased use of radiopharmaceuticals is further driving the growth of the market.
Nuclear Medicine Market – Global Forecast to 2030
“The hospitals segment is projected to account for the largest share during the forecast period.”
By end user, the hospitals segment is projected to account for the largest share of the nuclear medicine market during the forecast period. This projection is based on the capability of hospitals in preparing and administering radiopharmaceuticals through complex processes, available licensed facilities to deal with radioisotopes, and the presence of specialized nuclear medicine experts. Additionally, the presence of large volumes of patients, combined with diagnostic and therapeutic services, and well-established reimbursement systems in hospitals, is further expected to drive the growth of the segment in the coming years.
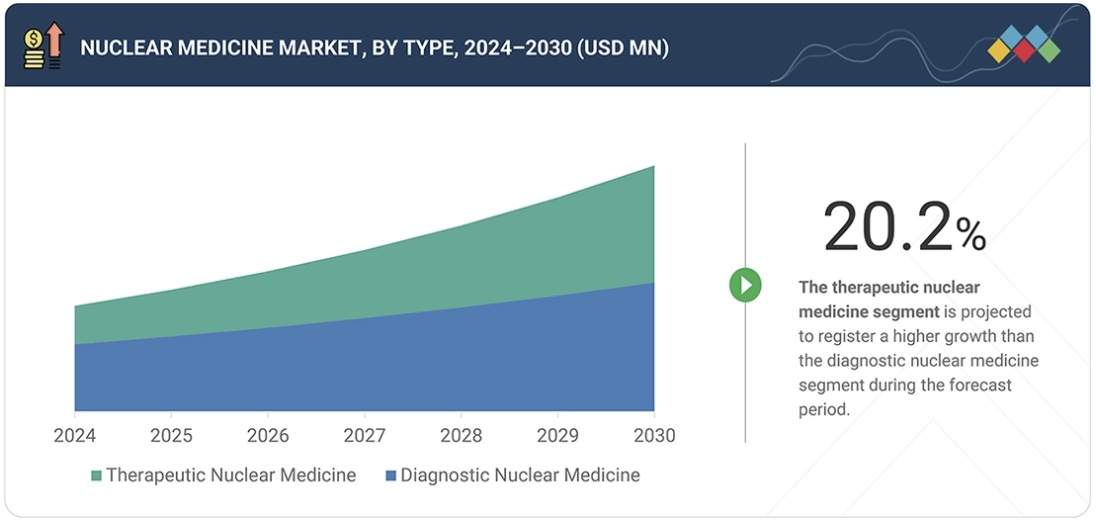
“The therapeutic applications segment is projected to register a higher growth than the diagnostic applications segment during the forecast period.”
The therapeutic applications segment is projected to register a higher growth than the diagnostics applications segment during the forecast period. This projection is driven by the rising application of targeted radiopharmaceutical therapies and radioisotopes in treating cancer, especially in theranostics. Increasing clinical efficacy of radionuclide therapies and growing regulatory approval and investment in the production of therapeutic radioisotopes are boosting the acceptance across healthcare providers.

Nuclear Medicine Market – Global Forecast to 2030 – region
“Asia Pacific is projected to witness the highest growth rate during the forecast period.”
Asia Pacific is projected to register the highest growth in the nuclear medicine market over the forecast period. This is due to the rising demand for radiopharmaceuticals and radioisotopes because of factors like increasing cancer prevalence, hospital facilities expansion, and growing access to nuclear medicine services. Additionally, the production of radioisotopes and the enhancement of regulatory infrastructure and supply chains by the governments are also increasing the growth of the markets in the region faster. Moreover, the growing healthcare spending and the rising awareness of early diagnosis and specific treatments are contributing to the rise in the number of nuclear medicine procedures.
In-depth interviews were conducted with chief executive officers (CEOs), directors, and other executives from various key organizations operating in the authentication and brand protection marketplace.
The breakdown of primary participants is mentioned below:
- By Company Type – Tier 1 – 64%; Tier 2 – 23%; and Tier 3 – 13%
- By Designation – C Level – 35%; Director Level – 25%; and Others – 40%
- By Region – North America – 44%; Europe – 23%; Asia Pacific – 28%; Latin America – 3%; Middle East & Africa – 2%
Key Players in the Nuclear Medicine Market
The key players operating in the nuclear medicine market include GE HealthCare (US), Cardinal Health (US), Curium (France), Bayer AG (Germany), Lantheus Holdings, Inc. (US), Novartis AG (Switzerland), Jubilant Pharmova Limited (India), Bracco Imaging S.P.A (Italy), Pharmalogic Holdings Corp. (US), NTP Radioisotopes SOC Ltd. (South Africa), Nordion Inc. (Canada), Siemens Healthineers AG (Germany), NorthStar Medical Radiosiotopes, LLC (US), Eckert & Ziegler (Germany), Isotope JSC (Russia), Global Medical Solutions (US), Telix Pharmaceuticals Limited (Australia), PDRadiopharma Inc. (Japan), ITM Isotope Technologies Munich SE (Germany), BWX Technologies Inc. (US), SHINE Technologies, LLC (US), Isotopia (Israel), Institutes of Isotopes (Hungary), China Isotope & Radiation Corporation (China), and IRE Elit (Turkey).

Nuclear Medicine Market – Global Forecast to 2030 – ecosystem
Research Coverage
The report analyzes the nuclear medicine market and aims to estimate the market size and future growth potential of various market segments by type, application, volume assessment, end user, and region. The report provides a competitive analysis of the key players operating in this market, along with their company profiles, product offerings, recent developments, and key market strategies.
Reasons to Buy the Report
This report will enrich established firms as well as new entrants/smaller firms to gauge the pulse of the market, which, in turn, would help them garner a greater share of the market. Firms purchasing the report could use one or a combination of the following strategies to strengthen their positions in the market.
This report provides insights into:
- Analysis of key drivers (Increasing incidence and prevalence of target conditions, development of alpha-radioimmunotherapy-based targeted cancer treatments, initiatives to reduce demand and supply gap of Mo-99), restraints (Short half-life of radiopharmaceuticals), opportunities (Use of radiopharmaceuticals in neurological applications, growth opportunities in emerging economies), challenges (Hospital budget cuts and high equipment costs) influencing the growth of the nuclear medicine market
- Product Development/Innovation: Detailed insights into upcoming technologies, research & development activities, and product launches in the nuclear medicine market
- Market Development: Comprehensive information on the lucrative emerging markets (based on type, application, volume assessment, and end user) across regions
- Market Diversification: Exhaustive information about the product portfolios, growing geographies, recent developments, and investments in the nuclear medicine market
- Competitive Assessment: In-depth assessment of market share, growth strategies, product offerings, and capabilities of the leading players [GE HealthCare (US), Cardinal Health (US), Curium (France), Bayer AG (Germany), and Lantheus Holdings, Inc. (US)] operating in the nuclear medicine market
Table of Contents
1 INTRODUCTION 41
1.1 STUDY OBJECTIVES 41
1.2 MARKET DEFINITION 41
1.3 STUDY SCOPE 42
1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE 42
1.3.2 REGIONAL SCOPE 43
1.3.3 INCLUSIONS & EXCLUSIONS 43
1.3.4 YEARS CONSIDERED 45
1.4 CURRENCY CONSIDERED 45
1.5 LIMITATIONS 46
1.6 STAKEHOLDERS 46
1.7 SUMMARY OF CHANGES 47
2 EXECUTIVE SUMMARY 49
2.1 MARKET HIGHLIGHTS & KEY INSIGHTS 49
2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS 51
2.3 DISRUPTIVE TRENDS IN NUCLEAR MEDICINE MARKET 52
2.4 HIGH-GROWTH SEGMENTS 53
2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST 54
3 PREMIUM INSIGHTS 55
3.1 NUCLEAR MEDICINE MARKET OVERVIEW 55
3.2 NORTH AMERICA: NUCLEAR MEDICINE MARKET, BY TYPE & COUNTRY 56
3.3 NUCLEAR MEDICINE MARKET: GEOGRAPHIC SNAPSHOT 56
3.4 NUCLEAR MEDICINE MARKET: DEVELOPED VS. EMERGING ECONOMIES 57
4 MARKET OVERVIEW 58
4.1 INTRODUCTION 58
4.2 MARKET DYNAMICS 59
4.2.1 DRIVERS 59
4.2.1.1 Increasing incidence and prevalence of target conditions 59
4.2.1.2 Rapid expansion of radioligand & targeted radionuclide therapy 60
4.2.1.3 Strategic entry of large pharma & capital inflows 61
4.2.1.4 Initiatives to reduce demand and supply gap of Mo-99 62
4.2.2 RESTRAINTS 64
4.2.2.1 Short half-life of radiopharmaceuticals 64
4.2.2.2 Structural isotope supply fragility 66
4.2.3 OPPORTUNITIES 67
4.2.3.1 Development of alpha-radioimmunotherapy-based targeted cancer treatments 67
4.2.3.2 Use of radiopharmaceuticals in neurological applications 68
4.2.3.3 Growth opportunities in emerging economies and expanding PET Infrastructure 69
4.2.4 CHALLENGES 71
4.2.4.1 High capital intensity of nuclear medicine infrastructure and hospital budget cuts 71
4.2.4.2 Workforce shortage in nuclear medicine 72
4.3 UNMET NEEDS & WHITE SPACES 73
4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES 73
4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS 74
5 INDUSTRY TRENDS 76
5.1 PORTER’S FIVE FORCES ANALYSIS 76
5.1.1 INTENSITY OF COMPETITIVE RIVALRY 77
5.1.2 BARGAINING POWER OF SUPPLIERS 77
5.1.3 BARGAINING POWER OF BUYERS 77
5.1.4 THREAT OF SUBSTITUTES 77
5.1.5 THREAT OF NEW ENTRANTS 78
5.2 MACROECONOMIC INDICATORS 78
5.2.1 INTRODUCTION 78
5.2.2 GDP TRENDS & FORECAST 78
5.2.3 TRENDS IN GLOBAL HEALTHCARE INDUSTRY 79
5.2.4 TRENDS IN GLOBAL PHARMACEUTICAL INDUSTRY 80
5.3 VALUE CHAIN ANALYSIS 80
5.4 ECOSYSTEM ANALYSIS 82
5.5 PRICING ANALYSIS 85
5.5.1 INDICATIVE PRICE OF NUCLEAR MEDICINE PRODUCTS, BY TYPE, 2024 85
5.5.2 INDICATIVE PRICE OF NUCLEAR MEDICINE PRODUCTS, BY REGION, 2024 86
5.6 TRADE ANALYSIS 87
5.6.1 IMPORT SCENARIO FOR HS CODE 2844 87
5.6.2 EXPORT SCENARIO FOR HS CODE 2844 88
5.6.3 IMPORT SCENARIO FOR HS CODE 3002 89
5.6.4 EXPORT SCENARIO FOR HS CODE 3002 89
5.7 KEY CONFERENCES & EVENTS, 2026–2027 90
5.8 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES 91
5.9 INVESTMENT & FUNDING SCENARIO 92
5.10 CASE STUDY ANALYSIS 93
5.11 IMPACT OF 2025 US TARIFFS ON NUCLEAR MEDICINE MARKET 95
5.11.1 INTRODUCTION 95
5.11.2 KEY TARIFF RATES 96
5.11.3 PRICE IMPACT ANALYSIS 97
5.11.4 IMPACT ON COUNTRY/REGION 97
5.11.4.1 US 97
5.11.4.2 Europe 97
5.11.4.3 Asia Pacific 98
5.11.5 IMPACT ON END-USE INDUSTRIES 98
5.11.5.1 Hospitals 98
5.11.5.2 Diagnostic & imaging centers 98
5.11.5.3 Research & academic institutes 99
6 STRATEGIC DISRUPTIONS THROUGH TECHNOLOGY, PATENTS, AND DIGITAL & AI ADOPTION 100
6.1 KEY EMERGING TECHNOLOGIES 100
6.1.1 NEXT-GENERATION RADIOLIGAND THERAPY & THERANOSTICS 100
6.1.2 RADIOISOTOPE PRODUCTION TECHNOLOGIES 100
6.1.3 HIGH-SPECIFIC-ACTIVITY RADIOLABELING AND CHELATION CHEMISTRY 101
6.1.4 GMP HOT-CELL MANUFACTURING & RADIOPHARMACEUTICAL QUALITY CONTROL 101
6.1.5 COLD KIT & GENERATOR TECHNOLOGIES 102
6.1.6 DECENTRALIZED ISOTOPE PRODUCTION & ADVANCED RADIOPHARMACY SYSTEMS 103
6.2 COMPLEMENTARY TECHNOLOGIES 103
6.2.1 AUTOMATED RADIOTRACER SYNTHESIS & DISPENSING SYSTEMS 103
6.2.2 DOSIMETRY & TREATMENT PLANNING TECHNOLOGIES 103
6.3 ADJACENT TECHNOLOGIES 104
6.3.1 TARGET DISCOVERY & RADIOLIGAND DESIGN PLATFORMS 104
6.3.2 INTEGRATED THERANOSTIC WORKFLOWS & DATA PLATFORMS 105
6.3.3 AI-ENABLED QUANTITATIVE IMAGING & WORKFLOW AUTOMATION 105
6.4 TECHNOLOGY/PRODUCT ROADMAP 105
6.5 PATENT ANALYSIS 106
6.5.1 PATENT PUBLICATION TRENDS FOR NUCLEAR MEDICINE MARKET 106
6.5.2 INSIGHTS: JURISDICTION & TOP APPLICANT ANALYSIS 107
6.6 FUTURE APPLICATIONS 109
6.6.1 PRECISION ONCOLOGY & THERANOSTICS EXPANSION 110
6.6.2 PERSONALIZED RADIONUCLIDE THERAPY USING DOSIMETRY-GUIDED DOSING 110
6.6.3 EARLY DISEASE DETECTION & RISK STRATIFICATION 111
6.6.4 NEURODEGENERATIVE & NEUROINFLAMMATORY DISORDERS 111
6.6.5 INFLAMMATION, INFECTION, AND IMMUNE SYSTEM IMAGING 111
6.7 IMPACT OF AI/GEN AI ON NUCLEAR MEDICINE MARKET 112
6.7.1 INTRODUCTION 112
6.7.2 MARKET POTENTIAL OF AI/GEN AI IN NUCLEAR MEDICINE MARKET 113
6.7.3 CASE STUDIES RELATED TO AI/GEN AI IMPLEMENTATION 115
6.7.3.1 Clinical validation of AI-enabled standardization and workflow efficiency in PSMA PET imaging for prostate cancer 115
6.7.4 IMPACT OF AI/GEN AI ON INTERCONNECTED & ADJACENT ECOSYSTEMS 116
6.7.4.1 Radiopharmacy, manufacturing, and quality operations 116
6.7.4.2 Clinical imaging, dosimetry, and treatment planning infrastructure 116
6.7.4.3 Hospital systems, regulatory compliance, and theranostic data platforms 117
6.7.5 USER READINESS AND IMPACT ASSESSMENT 117
6.7.5.1 User readiness 117
6.7.5.1.1 User A: Hospitals 117
6.7.5.1.2 User B: Diagnostic & imaging centers 117
6.7.5.2 Impact assessment 118
6.7.5.2.1 User A: Hospitals 118
6.7.5.2.1.1 Implementation 118
6.7.5.2.1.2 Impact 118
6.7.5.2.2 User B: Diagnostic & imaging centers 118
6.7.5.2.2.1 Implementation 118
6.7.5.2.2.2 Impact 118
7 REGULATORY LANDSCAPE 119
7.1 REGIONAL REGULATIONS & COMPLIANCE 119
7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 121
7.1.2 REGULATORY FRAMEWORK 127
7.1.2.1 North America 127
7.1.2.2 Europe 130
7.1.2.3 Asia Pacific 133
7.1.2.4 Latin America 135
7.1.2.5 Middle East & Africa 135
7.1.3 INDUSTRY STANDARDS 136
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR 138
8.1 INTRODUCTION 138
8.2 DECISION-MAKING PROCESS 138
8.3 BUYER STAKEHOLDERS & BUYING EVALUATION CRITERIA 139
8.3.1 KEY STAKEHOLDERS IN BUYING PROCESS 139
8.3.2 BUYING CRITERIA 140
8.4 ADOPTION BARRIERS & INTERNAL CHALLENGES 141
8.5 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES 142
8.5.1 UNMET NEEDS 142
8.5.2 END-USER EXPECTATIONS 144
8.6 MARKET PROFITABILITY 145
9 NUCLEAR MEDICINE MARKET, BY TYPE 146
9.1 INTRODUCTION 147
9.2 DIAGNOSTIC NUCLEAR MEDICINE 147
9.2.1 SPECT RADIOPHARMACEUTICALS 148
9.2.1.1 Tc-99m 151
9.2.1.1.1 Dominant role of Tc-99m in SPECT to boost market growth 151
9.2.1.2 I-123 152
9.2.1.2.1 Reduced exposure to radiation and lower risk of patients developing radiation-induced cancer to drive demand 152
9.2.1.3 Tl-201 153
9.2.1.3.1 Most commonly used substitute for Tc-99m in cardiac stress tests to support market growth 153
9.2.1.4 Ga-67 154
9.2.1.4.1 Long half-life of Ga-67 to facilitate easy sales and distribution 154
9.2.1.5 Other SPECT radiopharmaceuticals 155
9.2.2 PET RADIOPHARMACEUTICALS 156
9.2.2.1 F-18 157
9.2.2.1.1 Advantages such as highly accurate, metabolism-based cancer imaging to boost demand 157
9.2.2.2 Rb-82 158
9.2.2.2.1 High accuracy offered by Rb-82 isotopes to support market growth 158
9.2.2.3 Other PET radiopharmaceuticals 159
9.3 THERAPEUTIC NUCLEAR MEDICINE 160
9.3.1 ALPHA EMITTERS 161
9.3.1.1 Ra-223 162
9.3.1.1.1 Rising metastatic prostate cancer burden and demand for bone-targeted, outpatient alpha therapy to drive adoption 162
9.3.2 BETA EMITTERS 163
9.3.2.1 I-131 164
9.3.2.1.1 I-131 to account for largest share of beta emitters market during forecast period 164
9.3.2.2 Y-90 165
9.3.2.2.1 Rising incidence of liver cancers and hepatocellular carcinoma to propel growth 165
9.3.2.3 Sm-153 165
9.3.2.3.1 Increasing incidence of bone metastasis to drive market 165
9.3.2.4 Lu-177 166
9.3.2.4.1 Rising cancer burden and expanding PRRT adoption to fuel growth 166
9.3.2.5 Other beta emitters 167
9.3.3 BRACHYTHERAPY ISOTOPES 168
9.3.3.1 I-125 169
9.3.3.1.1 I-125 to account for largest share of brachytherapy isotopes market 169
9.3.3.2 Ir-192 170
9.3.3.2.1 Precise, high-dose brachytherapy across multiple cancers to propel market 170
9.3.3.3 Pd-103 171
9.3.3.3.1 Ability to make brachytherapy permanent implant seeds for early-stage prostate cancer to drive adoption 171
9.3.3.4 Cs-131 172
9.3.3.4.1 Growing use of Cs-131 in gynecological applications to aid growth 172
9.3.3.5 Other brachytherapy isotopes 172
10 NUCLEAR MEDICINE MARKET, BY APPLICATION 174
10.1 INTRODUCTION 175
10.2 DIAGNOSTIC APPLICATIONS 175
10.2.1 SPECT APPLICATIONS 176
10.2.1.1 Cardiology 178
10.2.1.1.1 Rising global cardiovascular disease burden and expanding, evidence-backed cardiology applications to drive growth 178
10.2.1.2 Bone scans 179
10.2.1.2.1 High precision of SPECT to boost adoption of bone scans 179
10.2.1.3 Thyroid 180
10.2.1.3.1 Rising demand for combination SPECT/CT therapy to drive market growth 180
10.2.1.4 Pulmonary scans 181
10.2.1.4.1 High accuracy and sensitivity of SPECT/CT pulmonary imaging to boost market 181
10.2.1.5 Other SPECT applications 182
10.2.2 PET APPLICATIONS 183
10.2.2.1 Oncology 184
10.2.2.1.1 Increasing incidence of cancer to fuel market growth 184
10.2.2.2 Cardiology 186
10.2.2.2.1 Growing preference for FDG in cardiac imaging to propel market 186
10.2.2.3 Neurology 187
10.2.2.3.1 Increasing incidence of AD, epilepsy, and Parkinson’s disease to increase adoption of PET imaging 187
10.2.2.4 Other PET applications 188
10.3 THERAPEUTIC APPLICATIONS 189
10.3.1 THYROID INDICATIONS 190
10.3.1.1 Increasing prevalence of thyroid disorders to boost market 190
10.3.2 BONE METASTASIS 191
10.3.2.1 Introduction of novel therapies for bone metastasis to positively impact market growth 191
10.3.3 ENDOCRINE TUMORS 192
10.3.3.1 US to dominate therapeutic nuclear medicine market for endocrine tumor applications 192
10.3.4 LYMPHOMA 193
10.3.4.1 Development of new isotopes for treatment of lymphoma to present huge growth opportunities 193
10.3.5 OTHER THERAPEUTIC APPLICATIONS 194
11 NUCLEAR MEDICINE MARKET: PROCEDURAL VOLUME ASSESSMENT 195
11.1 INTRODUCTION 196
11.2 DIAGNOSTIC PROCEDURES 197
11.2.1 HIGH PREVALENCE OF CANCER AND CARDIAC DISEASES TO DRIVE MARKET GROWTH 197
11.3 THERAPEUTIC PROCEDURES 198
11.3.1 GROWING DEMAND FOR NON-INVASIVE METHODS TO SUPPORT MARKET GROWTH 198
12 NUCLEAR MEDICINE MARKET, BY END USER 200
12.1 INTRODUCTION 201
12.2 HOSPITALS 202
12.2.1 GROWING VOLUME OF DIAGNOSTIC PROCEDURES TO DRIVE MARKET GROWTH 202
12.3 DIAGNOSTIC & IMAGING CENTERS 203
12.3.1 GROWING NUMBER OF PRIVATE IMAGING CENTERS TO SUPPORT MARKET GROWTH 203
12.4 ACADEMIC & RESEARCH INSTITUTES 204
12.4.1 INCREASING COLLABORATIONS BETWEEN NUCLEAR IMAGING COMPANIES AND ACADEMIA TO PROPEL MARKET GROWTH 204
12.5 OTHER END USERS 206
13 NUCLEAR MEDICINE MARKET, BY REGION 207
13.1 INTRODUCTION 208
13.2 NORTH AMERICA 209
13.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA 210
13.2.2 US 218
13.2.2.1 US to dominate North American nuclear medicine market during forecast period 218
13.2.3 CANADA 226
13.2.3.1 Increasing initiatives for medical isotope development to support market growth 226
13.3 EUROPE 233
13.3.1 MACROECONOMIC OUTLOOK FOR EUROPE 234
13.3.2 GERMANY 242
13.3.2.1 Well-established healthcare system to drive market growth 242
13.3.3 FRANCE 250
13.3.3.1 Rising incidence of diseases such as cancer, Alzheimer’s, and Parkinson’s disease to favor market growth 250
13.3.4 UK 258
13.3.4.1 Growing demand for diagnostic imaging and growing awareness to drive market growth 258
13.3.5 ITALY 265
13.3.5.1 Aging demographics, high procedure volumes, increasing cancer incidence, and domestic isotope self-sufficiency to fuel growth 265
13.3.6 SPAIN 273
13.3.6.1 Rising disease prevalence, regulatory modernization, and targeted radioligand expansion to create supportive environment 273
13.3.7 REST OF EUROPE 280
13.4 ASIA PACIFIC 288
13.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC 288
13.4.2 JAPAN 298
13.4.2.1 Japan to dominate APAC nuclear medicine market 298
13.4.3 CHINA 306
13.4.3.1 Increasing nuclear medicine infrastructure and growing prevalence of chronic diseases to augment market 306
13.4.4 INDIA 314
13.4.4.1 Rising cancer and cardiovascular disease burden and rapid expansion of nuclear medicine infrastructure to boost market 314
13.4.5 SOUTH KOREA 321
13.4.5.1 Accelerating PET utilization and maturing domestic RPT innovation ecosystem to augment market 321
13.4.6 AUSTRALIA 328
13.4.6.1 Rising cancer prevalence, high PET penetration, and growing domestic isotope production to support growth 328
13.4.7 REST OF ASIA PACIFIC 335
13.5 LATIN AMERICA 342
13.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA 342
13.5.2 BRAZIL 349
13.5.2.1 Rising cancer incidence and expanding imaging infrastructure to drive market 349
13.5.3 MEXICO 356
13.5.3.1 Rising cancer and cardiovascular disease burden and expanding private-sector PET access to drive market 356
13.5.4 REST OF LATIN AMERICA 362
13.6 MIDDLE EAST & AFRICA 368
13.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA 368
13.6.2 GCC COUNTRIES 375
13.6.2.1 Growing healthcare needs and favorable government support to boost market 375
13.6.3 REST OF MIDDLE EAST & AFRICA 380
14 COMPETITIVE LANDSCAPE 387
14.1 OVERVIEW 387
14.2 KEY PLAYER STRATEGIES/RIGHT TO WIN 387
14.2.1 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN NUCLEAR MEDICINE MARKET 388
14.3 REVENUE ANALYSIS, 2020–2024 391
14.4 MARKET SHARE ANALYSIS, 2024 392
14.5 BRAND/PRODUCT COMPARISON 396
14.6 COMPANY VALUATION & FINANCIAL METRICS 397
14.6.1 FINANCIAL METRICS 397
14.6.2 COMPANY VALUATION 397
14.7 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2024 398
14.7.1 STARS 398
14.7.2 EMERGING LEADERS 398
14.7.3 PERVASIVE PLAYERS 398
14.7.4 PARTICIPANTS 399
14.7.5 COMPANY FOOTPRINT: KEY PLAYERS, 2024 400
14.7.5.1 Company footprint 400
14.7.5.2 Region footprint 401
14.7.5.3 Type footprint 402
14.7.5.4 Application footprint 403
14.8 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2024 404
14.8.1 PROGRESSIVE COMPANIES 404
14.8.2 RESPONSIVE COMPANIES 404
14.8.3 DYNAMIC COMPANIES 404
14.8.4 STARTING BLOCKS 404
14.8.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2024 406
14.8.5.1 Detailed list of key startups/SMEs 406
14.8.5.2 Competitive benchmarking of startups/SMEs 406
14.9 COMPETITIVE SCENARIO 408
14.9.1 PRODUCT APPROVALS 408
14.9.2 DEALS 410
14.9.3 EXPANSIONS 412
14.9.4 OTHER DEVELOPMENTS 413
15 COMPANY PROFILES 414
15.1 KEY PLAYERS 414
15.1.1 NOVARTIS AG 414
15.1.1.1 Business overview 414
15.1.1.2 Products offered 415
15.1.1.3 Recent developments 416
15.1.1.3.1 Product approvals 416
15.1.1.3.2 Deals 417
15.1.1.3.3 Expansions 418
15.1.1.4 MnM view 419
15.1.1.4.1 Right to win 419
15.1.1.4.2 Strategic choices 420
15.1.1.4.3 Weaknesses & competitive threats 420
15.1.2 LANTHEUS HOLDINGS, INC. 421
15.1.2.1 Business overview 421
15.1.2.2 Products offered 422
15.1.2.3 Recent developments 423
15.1.2.3.1 Deals 423
15.1.2.4 MnM view 424
15.1.2.4.1 Right to win 424
15.1.2.4.2 Strategic choices 424
15.1.2.4.3 Weaknesses & competitive threats 424
15.1.3 GE HEALTHCARE 425
15.1.3.1 Business overview 425
15.1.3.2 Products offered 426
15.1.3.3 Recent developments 427
15.1.3.3.1 Product approvals 427
15.1.3.3.2 Deals 427
15.1.3.3.3 Expansions 429
15.1.3.3.4 Other developments 429
15.1.3.4 MnM view 430
15.1.3.4.1 Right to win 430
15.1.3.4.2 Strategic choices 430
15.1.3.4.3 Weaknesses & competitive threats 430
15.1.4 CURIUM 431
15.1.4.1 Business overview 431
15.1.4.2 Products offered 431
15.1.4.3 Recent developments 435
15.1.4.3.1 Product approvals & enhancements 435
15.1.4.3.2 Deals 436
15.1.4.3.3 Expansions 438
15.1.4.3.4 Other developments 438
15.1.4.4 MnM view 438
15.1.4.4.1 Right to win 438
15.1.4.4.2 Strategic choices 438
15.1.4.4.3 Weaknesses & competitive threats 439
15.1.5 TELIX PHARMACEUTICALS LIMITED 440
15.1.5.1 Business overview 440
15.1.5.2 Products offered 441
15.1.5.3 Recent developments 442
15.1.5.3.1 Product approvals 442
15.1.5.3.2 Deals 443
15.1.5.3.3 Other developments 444
15.1.5.4 MnM view 444
15.1.5.4.1 Right to win 444
15.1.5.4.2 Strategic choices 444
15.1.5.4.3 Weaknesses & competitive threats 445
15.1.6 SIEMENS HEALTHINEERS 446
15.1.6.1 Business overview 446
15.1.6.2 Products offered 447
15.1.6.3 Recent developments 448
15.1.6.3.1 Deals 448
15.1.6.3.2 Other developments 448
15.1.7 CHINA ISOTOPE & RADIATION CORPORATION 449
15.1.7.1 Business overview 449
15.1.7.2 Products offered 450
15.1.7.3 Recent developments 451
15.1.7.3.1 Other developments 451
15.1.8 BAYER AG 452
15.1.8.1 Business overview 452
15.1.8.2 Products offered 453
15.1.8.3 Recent developments 454
15.1.8.3.1 Deals 454
15.1.9 BRACCO IMAGING S.P.A. 455
15.1.9.1 Business overview 455
15.1.9.2 Products offered 455
15.1.9.3 Recent developments 456
15.1.9.3.1 Product approvals 456
15.1.9.3.2 Deals 456
15.1.9.3.3 Other developments 457
15.1.10 CARDINAL HEALTH 458
15.1.10.1 Business overview 458
15.1.10.2 Products offered 459
15.1.10.3 Recent developments 462
15.1.10.3.1 Deals 462
15.1.11 JUBILANT PHARMOVA LIMITED 463
15.1.11.1 Business overview 463
15.1.11.2 Products offered 464
15.1.11.3 Recent developments 466
15.1.11.3.1 Product approvals 466
15.1.11.3.2 Deals 466
15.1.11.3.3 Other developments 467
15.1.12 ELI LILLY AND COMPANY 468
15.1.12.1 Business overview 468
15.1.12.2 Products offered 469
15.1.12.3 Recent developments 470
15.1.12.3.1 Deals 470
15.1.12.3.2 Other developments 471
15.1.13 BWXT MEDICAL LTD. 472
15.1.13.1 Business overview 472
15.1.13.2 Products offered 472
15.1.13.3 Recent developments 473
15.1.13.3.1 Product approvals 473
15.1.14 NTP RADIOISOTOPES SOC LTD. (A SUBSIDIARY OF SOUTH AFRICAN NUCLEAR ENERGY CORPORATION) 474
15.1.14.1 Business overview 474
15.1.14.2 Products offered 474
15.1.14.3 Recent developments 475
15.1.14.3.1 Deals 475
15.1.15 ECKERT & ZIEGLER 476
15.1.15.1 Business overview 476
15.1.15.2 Products offered 477
15.1.15.3 Recent developments 478
15.1.15.3.1 Product approvals 478
15.1.15.3.2 Deals 478
15.1.15.3.3 Expansions 480
15.1.16 ISOTOPE JSC 481
15.1.16.1 Business overview 481
15.1.16.2 Products offered 481
15.1.16.3 Recent developments 482
15.1.16.3.1 Deals 482
15.1.16.3.2 Other developments 483
15.1.17 PDRADIOPHARMA INC. 484
15.1.17.1 Business overview 484
15.1.17.2 Products offered 484
15.1.17.3 Recent developments 486
15.1.17.3.1 Product launches & approvals 486
15.1.17.3.2 Deals 487
15.1.17.3.3 Expansions 487
15.1.18 ITM ISOTOPE TECHNOLOGIES MUNICH SE 488
15.1.18.1 Business overview 488
15.1.18.2 Products offered 489
15.1.18.3 Recent developments 489
15.1.18.3.1 Product approvals 489
15.1.18.3.2 Deals 490
15.1.18.3.3 Expansions 491
15.1.18.3.4 Other developments 492
15.1.19 NARODOWE CENTRUM BADAŃ JĄDROWYCH OŚRODEK RADIOIZOTOPÓW – POLATOM 493
15.1.19.1 Business overview 493
15.1.19.2 Products offered 494
15.1.19.3 Recent developments 496
15.1.19.3.1 Deals 496
15.1.20 ANSTO 497
15.1.20.1 Business overview 497
15.1.20.2 Products offered 498
15.1.20.3 Recent developments 499
15.1.20.3.1 Deals 499
15.1.20.3.2 Other developments 499
15.2 OTHER PLAYERS 500
15.2.1 SHINE TECHNOLOGIES, LLC 500
15.2.2 ISOTOPIA MOLECULAR IMAGING LTD. 501
15.2.3 INSTITUTE OF ISOTOPES 502
15.2.4 GLOBAL MEDICAL SOLUTIONS 504
15.2.5 ISOTEX DIAGNOSTICS 505
16 RESEARCH METHODOLOGY 506
16.1 RESEARCH DATA 506
16.2 RESEARCH APPROACH 506
16.2.1 SECONDARY RESEARCH 507
16.2.1.1 Key data from secondary sources 509
16.2.2 PRIMARY RESEARCH 509
16.2.2.1 Primary sources 510
16.2.2.2 Key data from primary sources 511
16.2.2.3 Breakdown of primaries 511
16.2.2.4 Insights from primary experts 512
16.3 RESEARCH METHODOLOGY DESIGN 512
16.4 MARKET SIZE ESTIMATION 514
16.4.1 APPROACH 1: REVENUE SHARE ANALYSIS (BOTTOM-UP APPROACH – SUPPLY-SIDE ANALYSIS) 514
16.4.2 APPROACH 2: SECONDARY DATA & PRIMARY INTERVIEWS 517
16.4.3 APPROACH 3: SEGMENTAL MARKET SIZE ASSESSMENT 517
16.4.4 APPROACH 4: TOP-DOWN APPROACH 518
16.4.5 GROWTH FORECAST 518
16.4.6 GEOGRAPHIC MARKET ASSESSMENT (BY REGION AND COUNTRY) 520
16.5 MARKET BREAKDOWN & DATA TRIANGULATION 521
16.6 STUDY ASSUMPTIONS 522
16.7 RESEARCH LIMITATIONS 523
16.7.1 METHODOLOGY-RELATED LIMITATIONS 523
16.7.2 SCOPE-RELATED LIMITATIONS 523
16.8 RISK ASSESSMENT 523
17 APPENDIX 524
17.1 DISCUSSION GUIDE 524
17.2 KNOWLEDGESTORE: MARKETSANDMARKETS’ SUBSCRIPTION PORTAL 532
17.3 CUSTOMIZATION OPTIONS 534
17.4 RELATED REPORTS 534
17.5 AUTHOR DETAILS 535
LIST OF TABLES
TABLE 1 EXCHANGE RATES UTILIZED FOR CONVERSION TO USD 46
TABLE 2 MAIN MO-99 PRODUCTION REACTORS 63
TABLE 3 RADIOPHARMACEUTICALS: PHYSICAL HALF-LIFE AND MAJOR CLINICAL APPLICATIONS 64
TABLE 4 UK: NUCLEAR MEDICINE-RELATED WORKFORCE AND VACANCY DATA, 2021 72
TABLE 5 NUCLEAR MEDICINE MARKET: PORTER’S FIVE FORCES ANALYSIS 77
TABLE 6 NUCLEAR MEDICINE MARKET: ROLE IN ECOSYSTEM 84
TABLE 7 INDICATIVE PRICE OF NUCLEAR MEDICINE PRODUCTS, BY TYPE, 2024 85
TABLE 8 INDICATIVE PRICE OF NUCLEAR MEDICINE IN EASTERN EUROPE, BY TYPE, 2024 86
TABLE 9 INDICATIVE PRICE OF NUCLEAR MEDICINE IN MENAT REGION, BY TYPE, 2024 86
TABLE 10 AVERAGE SELLING PRICE OF RADIOPHARMACEUTICALS IN INDIA, BY TYPE, 2024 87
TABLE 11 IMPORT VALUE FOR HS CODE 2844, 2020–2024 (USD THOUSAND) 87
TABLE 12 EXPORT VALUE FOR HS CODE 2844, 2020–2024 (USD THOUSAND) 88
TABLE 13 IMPORT VALUE FOR HS CODE 3002, 2020–2024 (USD THOUSAND) 89
TABLE 14 EXPORT VALUE FOR HS CODE 3002, 2020–2024 (USD THOUSAND) 89
TABLE 15 NUCLEAR MEDICINE MARKET: KEY CONFERENCES & EVENTS, 2026–2027 90
TABLE 16 CASE 1: BETA-AMYLOID PET/CT IMAGING ENABLES ALZHEIMER’S PATIENT QUALIFICATION FOR ANTI-AMYLOID THERAPY AT NEW YORK-PRESBYTERIAN HOSPITAL 93
TABLE 17 CASE 2: WHOLE-BODY PET/CT WITH EXTENDED AXIAL FOV ENABLES DETECTION OF RARE PERICARDIAL RECURRENCE IN PLEURAL MESOTHELIOMA AT RIGSHOSPITALET 93
TABLE 18 CASE 3: CLINICAL AND OPERATIONAL IMPACT OF IMPLEMENTING AN INTEGRATED THERANOSTICS PROGRAM IN BRAZIL 94
TABLE 19 US-ADJUSTED RECIPROCAL TARIFF RATES 96
TABLE 20 JURISDICTION ANALYSIS OF TOP APPLICANT COUNTRIES FOR NUCLEAR MEDICINE MARKET 108
TABLE 21 NUCLEAR MEDICINE MARKET: LIST OF PATENTS/PATENT APPLICATIONS 109
TABLE 22 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 121
TABLE 23 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 122
TABLE 24 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 124
TABLE 25 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 125
TABLE 26 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS 126
TABLE 27 REGULATORY SCENARIO OF NORTH AMERICA 127
TABLE 28 REGULATORY SCENARIO OF EUROPE 130
TABLE 29 REGULATORY SCENARIO OF ASIA PACIFIC 133
TABLE 30 REGULATORY SCENARIO OF LATIN AMERICA 135
TABLE 31 REGULATORY SCENARIO OF MIDDLE EAST & AFRICA 135
TABLE 32 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS OF TOP THREE
END USERS (%) 140
TABLE 33 KEY BUYING CRITERIA FOR TOP THREE END USERS 141
TABLE 34 UNMET NEEDS IN NUCLEAR MEDICINE MARKET 143
TABLE 35 END-USER EXPECTATIONS IN NUCLEAR MEDICINE MARKET 144
TABLE 36 NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 147
TABLE 37 DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 148
TABLE 38 DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY REGION,
2023–2030 (USD MILLION) 148
TABLE 39 ALTERNATIVES TO COMMON TC-99M-BASED DIAGNOSTIC PROCEDURES DUE TO SEVERE TC-99M SHORTAGES 149
TABLE 40 SPECT RADIOPHARMACEUTICALS MARKET, BY TYPE, 2023–2030 (USD MILLION) 151
TABLE 41 SPECT RADIOPHARMACEUTICALS MARKET, BY REGION,
2023–2030 (USD MILLION) 151
TABLE 42 TC-99M MARKET, BY REGION, 2023–2030 (USD MILLION) 152
TABLE 43 I-123 MARKET, BY REGION, 2023–2030 (USD MILLION) 153
TABLE 44 TI-201 MARKET, BY REGION, 2023–2030 (USD MILLION) 154
TABLE 45 GA-67 MARKET, BY REGION, 2023–2030 (USD MILLION) 155
TABLE 46 OTHER SPECT RADIOPHARMACEUTICALS MARKET, BY REGION,
2023–2030 (USD MILLION) 155
TABLE 47 PET RADIOPHARMACEUTICALS MARKET, BY TYPE, 2023–2030 (USD MILLION) 157
TABLE 48 PET RADIOPHARMACEUTICALS MARKET, BY REGION, 2023–2030 (USD MILLION) 157
TABLE 49 F-18 MARKET, BY REGION, 2023–2030 (USD MILLION) 158
TABLE 50 RB-82 MARKET, BY REGION, 2023–2030 (USD MILLION) 159
TABLE 51 OTHER PET RADIOPHARMACEUTICALS MARKET, BY REGION,
2023–2030 (USD MILLION) 160
TABLE 52 THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 161
TABLE 53 THERAPEUTIC NUCLEAR MEDICINE MARKET, BY REGION,
2023–2030 (USD MILLION) 161
TABLE 54 ALPHA EMITTERS MARKET, BY TYPE, 2023–2030 (USD MILLION) 162
TABLE 55 RA-223 MARKET, BY REGION, 2023–2030 (USD MILLION) 163
TABLE 56 BETA EMITTERS MARKET, BY TYPE, 2023–2030 (USD MILLION) 163
TABLE 57 BETA EMITTERS MARKET, BY REGION, 2023–2030 (USD MILLION) 164
TABLE 58 I-131 MARKET, BY REGION, 2023–2030 (USD MILLION) 164
TABLE 59 Y-90 MARKET, BY REGION, 2023–2030 (USD MILLION) 165
TABLE 60 SM-153 MARKET, BY REGION, 2023–2030 (USD MILLION) 166
TABLE 61 LU-177 MARKET, BY REGION, 2023–2030 (USD MILLION) 167
TABLE 62 OTHER BETA EMITTERS MARKET, BY REGION, 2023–2030 (USD MILLION) 168
TABLE 63 BRACHYTHERAPY ISOTOPES MARKET, BY TYPE, 2023–2030 (USD MILLION) 169
TABLE 64 BRACHYTHERAPY ISOTOPES MARKET, BY REGION, 2023–2030 (USD MILLION) 169
TABLE 65 I-125 MARKET, BY REGION, 2023–2030 (USD MILLION) 170
TABLE 66 IR-192 MARKET, BY REGION, 2023–2030 (USD MILLION) 171
TABLE 67 PD-103 MARKET, BY REGION, 2023–2030 (USD MILLION) 171
TABLE 68 CS-131 MARKET, BY REGION, 2023–2030 (USD MILLION) 172
TABLE 69 OTHER BRACHYTHERAPY ISOTOPES MARKET, BY REGION,
2023–2030 (USD MILLION) 173
TABLE 70 NUCLEAR MEDICINE MARKET, BY APPLICATION, 2023–2030 (USD MILLION) 175
TABLE 71 NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 176
TABLE 72 NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY REGION,
2023–2030 (USD MILLION) 176
TABLE 73 NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 177
TABLE 74 NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY REGION,
2023–2030 (USD MILLION) 177
TABLE 75 NUCLEAR MEDICINE MARKET FOR SPECT CARDIOLOGY APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 179
TABLE 76 NUCLEAR MEDICINE MARKET FOR SPECT BONE SCAN APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 180
TABLE 77 NUCLEAR MEDICINE MARKET FOR SPECT THYROID APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 181
TABLE 78 NUCLEAR MEDICINE MARKET FOR SPECT PULMONARY SCAN APPLICATIONS,
BY REGION, 2023–2030 (USD MILLION) 182
TABLE 79 NUCLEAR MEDICINE MARKET FOR OTHER SPECT APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 183
TABLE 80 NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 184
TABLE 81 NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY REGION,
2023–2030 (USD MILLION) 184
TABLE 82 NUCLEAR MEDICINE MARKET FOR PET ONCOLOGY APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 186
TABLE 83 NUCLEAR MEDICINE MARKET FOR PET CARDIOLOGY APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 187
TABLE 84 NUCLEAR MEDICINE MARKET FOR PET NEUROLOGY APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 188
TABLE 85 NUCLEAR MEDICINE MARKET FOR OTHER PET APPLICATIONS, BY REGION,
2023–2030 (USD MILLION) 188
TABLE 86 NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 190
TABLE 87 NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 190
TABLE 88 NUCLEAR MEDICINE MARKET FOR THYROID INDICATIONS, BY REGION,
2023–2030 (USD MILLION) 191
TABLE 89 NUCLEAR MEDICINE MARKET FOR BONE METASTASIS APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 192
TABLE 90 NUCLEAR MEDICINE MARKET FOR ENDOCRINE TUMOR APPLICATIONS, BY REGION, 2023–2030 (USD MILLION) 193
TABLE 91 NUCLEAR MEDICINE MARKET FOR LYMPHOMA APPLICATIONS, BY REGION,
2023–2030 (USD MILLION) 194
TABLE 92 NUCLEAR MEDICINE MARKET FOR OTHER THERAPEUTIC APPLICATIONS,
BY REGION, 2023–2030 (USD MILLION) 194
TABLE 93 NUCLEAR MEDICINE MARKET, BY PROCEDURE,
2023–2030 (THOUSAND PROCEDURES) 196
TABLE 94 NUCLEAR MEDICINE MARKET, BY REGION,
2023–2030 (THOUSAND PROCEDURES) 196
TABLE 95 NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC PROCEDURES, BY TYPE,
2023–2030 (THOUSAND PROCEDURES) 197
TABLE 96 NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC PROCEDURES, BY REGION,
2023–2030 (THOUSAND PROCEDURES) 198
TABLE 97 NUCLEAR MEDICINE MARKET FOR THERAPEUTIC PROCEDURES, BY REGION, 2023–2030 (THOUSAND PROCEDURES) 199
TABLE 98 NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 201
TABLE 99 NUCLEAR MEDICINE MARKET FOR HOSPITALS, BY REGION,
2023–2030 (USD MILLION) 203
TABLE 100 NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC & IMAGING CENTERS, BY REGION, 2023–2030 (USD MILLION) 204
TABLE 101 NUCLEAR MEDICINE MARKET FOR ACADEMIC & RESEARCH INSTITUTES,
BY REGION, 2023–2030 (USD MILLION) 205
TABLE 102 NUCLEAR MEDICINE MARKET FOR OTHER END USERS, BY REGION,
2023–2030 (USD MILLION) 206
TABLE 103 NUCLEAR MEDICINE MARKET, BY REGION, 2023–2030 (USD MILLION) 209
TABLE 104 NORTH AMERICA: KEY MACROECONOMIC INDICATORS 211
TABLE 105 NORTH AMERICA: NUCLEAR MEDICINE MARKET, BY COUNTRY,
2023–2030 (USD MILLION) 212
TABLE 106 NORTH AMERICA: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 212
TABLE 107 NORTH AMERICA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 212
TABLE 108 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 213
TABLE 109 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 213
TABLE 110 NORTH AMERICA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 214
TABLE 111 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE, 2023–2030 (USD MILLION) 214
TABLE 112 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 215
TABLE 113 NORTH AMERICA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 215
TABLE 114 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 216
TABLE 115 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 216
TABLE 116 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 217
TABLE 117 NORTH AMERICA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 217
TABLE 118 NORTH AMERICA: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 218
TABLE 119 US: CANCER INCIDENCE, BY CANCER TYPE, 2020 VS. 2040 220
TABLE 120 US: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 220
TABLE 121 US: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 220
TABLE 122 US: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 221
TABLE 123 US: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 221
TABLE 124 US: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 222
TABLE 125 US: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 222
TABLE 126 US: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 223
TABLE 127 US: NUCLEAR MEDICINE MARKET, BY APPLICATION, 2023–2030 (USD MILLION) 223
TABLE 128 US: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 223
TABLE 129 US: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 224
TABLE 130 US: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 224
TABLE 131 US: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 225
TABLE 132 US: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 225
TABLE 133 CANADA: CANCER INCIDENCE, BY CANCER TYPE, 2020 VS. 2040 227
TABLE 134 CANADA: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 227
TABLE 135 CANADA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 227
TABLE 136 CANADA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 228
TABLE 137 CANADA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 228
TABLE 138 CANADA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 229
TABLE 139 CANADA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 229
TABLE 140 CANADA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 230
TABLE 141 CANADA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 230
TABLE 142 CANADA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 231
TABLE 143 CANADA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 231
TABLE 144 CANADA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 232
TABLE 145 CANADA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 232
TABLE 146 CANADA: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 233
TABLE 147 EUROPE: KEY MACROECONOMIC INDICATORS 235
TABLE 148 EUROPE: NUCLEAR MEDICINE MARKET, BY COUNTRY, 2023–2030 (USD MILLION) 235
TABLE 149 EUROPE: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 236
TABLE 150 EUROPE: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 236
TABLE 151 EUROPE: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 237
TABLE 152 EUROPE: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 237
TABLE 153 EUROPE: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 238
TABLE 154 EUROPE: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 238
TABLE 155 EUROPE: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 239
TABLE 156 EUROPE: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 239
TABLE 157 EUROPE: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 240
TABLE 158 EUROPE: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 240
TABLE 159 EUROPE: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 241
TABLE 160 EUROPE: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 241
TABLE 161 EUROPE: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 242
TABLE 162 GERMANY: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 243
TABLE 163 GERMANY: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 244
TABLE 164 GERMANY: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 244
TABLE 165 GERMANY: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 245
TABLE 166 GERMANY: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 245
TABLE 167 GERMANY: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 246
TABLE 168 GERMANY: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES,
BY TYPE, 2023–2030 (USD MILLION) 246
TABLE 169 GERMANY: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 247
TABLE 170 GERMANY: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 247
TABLE 171 GERMANY: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 248
TABLE 172 GERMANY: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 248
TABLE 173 GERMANY: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 249
TABLE 174 GERMANY: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 249
TABLE 175 FRANCE: CANCER INCIDENCE, BY CANCER TYPE, 2020 VS. 2040 251
TABLE 176 FRANCE: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 251
TABLE 177 FRANCE: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 252
TABLE 178 FRANCE: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 252
TABLE 179 FRANCE: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 253
TABLE 180 FRANCE: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 253
TABLE 181 FRANCE: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 254
TABLE 182 FRANCE: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 254
TABLE 183 FRANCE: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 255
TABLE 184 FRANCE: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 255
TABLE 185 FRANCE: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 256
TABLE 186 FRANCE: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 256
TABLE 187 FRANCE: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 257
TABLE 188 FRANCE: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 257
TABLE 189 UK: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 259
TABLE 190 UK: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 259
TABLE 191 UK: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 260
TABLE 192 UK: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 260
TABLE 193 UK: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 261
TABLE 194 UK: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 261
TABLE 195 UK: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 262
TABLE 196 UK: NUCLEAR MEDICINE MARKET, BY APPLICATION, 2023–2030 (USD MILLION) 262
TABLE 197 UK: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 263
TABLE 198 UK: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 263
TABLE 199 UK: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 264
TABLE 200 UK: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 264
TABLE 201 UK: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 265
TABLE 202 ITALY: CANCER INCIDENCE, BY CANCER TYPE, 2020 VS. 2040 266
TABLE 203 ITALY: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 266
TABLE 204 ITALY: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 267
TABLE 205 ITALY: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 267
TABLE 206 ITALY: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 268
TABLE 207 ITALY: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 268
TABLE 208 ITALY: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 269
TABLE 209 ITALY: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 269
TABLE 210 ITALY: NUCLEAR MEDICINE MARKET, BY APPLICATION, 2023–2030 (USD MILLION) 270
TABLE 211 ITALY: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 270
TABLE 212 ITALY: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 271
TABLE 213 ITALY: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 271
TABLE 214 ITALY: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 272
TABLE 215 ITALY: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 272
TABLE 216 SPAIN: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 274
TABLE 217 SPAIN: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 274
TABLE 218 SPAIN: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 275
TABLE 219 SPAIN: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 275
TABLE 220 SPAIN: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 276
TABLE 221 SPAIN: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 276
TABLE 222 SPAIN: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 277
TABLE 223 SPAIN: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 277
TABLE 224 SPAIN: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 278
TABLE 225 SPAIN: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 278
TABLE 226 SPAIN: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 279
TABLE 227 SPAIN: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 279
TABLE 228 SPAIN: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 280
TABLE 229 REST OF EUROPE: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 282
TABLE 230 REST OF EUROPE: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 282
TABLE 231 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 283
TABLE 232 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 283
TABLE 233 REST OF EUROPE: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 284
TABLE 234 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE, 2023–2030 (USD MILLION) 284
TABLE 235 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 285
TABLE 236 REST OF EUROPE: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 285
TABLE 237 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 286
TABLE 238 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 286
TABLE 239 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 287
TABLE 240 REST OF EUROPE: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 287
TABLE 241 REST OF EUROPE: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 288
TABLE 242 ASIA PACIFIC: KEY MACROECONOMIC INDICATORS 289
TABLE 243 ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY COUNTRY,
2023–2030 (USD MILLION) 291
TABLE 244 ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 291
TABLE 245 ASIA PACIFIC: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 292
TABLE 246 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 292
TABLE 247 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 293
TABLE 248 ASIA PACIFIC: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 293
TABLE 249 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 294
TABLE 250 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES,
BY TYPE, 2023–2030 (USD MILLION) 294
TABLE 251 ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 295
TABLE 252 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 295
TABLE 253 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 296
TABLE 254 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 296
TABLE 255 ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 297
TABLE 256 ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 297
TABLE 257 JAPAN: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 299
TABLE 258 JAPAN: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 300
TABLE 259 JAPAN: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 300
TABLE 260 JAPAN: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 301
TABLE 261 JAPAN: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 301
TABLE 262 JAPAN: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 302
TABLE 263 JAPAN: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 302
TABLE 264 JAPAN: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 303
TABLE 265 JAPAN: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 303
TABLE 266 JAPAN: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 304
TABLE 267 JAPAN: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 304
TABLE 268 JAPAN: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 305
TABLE 269 JAPAN: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 305
TABLE 270 CHINA: CANCER INCIDENCE, BY CANCER TYPE, 2020 VS. 2040 307
TABLE 271 CHINA: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 307
TABLE 272 CHINA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 308
TABLE 273 CHINA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 308
TABLE 274 CHINA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 309
TABLE 275 CHINA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 309
TABLE 276 CHINA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 310
TABLE 277 CHINA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 310
TABLE 278 CHINA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 311
TABLE 279 CHINA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 311
TABLE 280 CHINA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 312
TABLE 281 CHINA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 312
TABLE 282 CHINA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 313
TABLE 283 CHINA: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 313
TABLE 284 INDIA: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 315
TABLE 285 INDIA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 315
TABLE 286 INDIA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 316
TABLE 287 INDIA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 316
TABLE 288 INDIA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 317
TABLE 289 INDIA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 317
TABLE 290 INDIA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 318
TABLE 291 INDIA: NUCLEAR MEDICINE MARKET, BY APPLICATION, 2023–2030 (USD MILLION) 318
TABLE 292 INDIA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 319
TABLE 293 INDIA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 319
TABLE 294 INDIA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 320
TABLE 295 INDIA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 320
TABLE 296 INDIA: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 321
TABLE 297 SOUTH KOREA: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 322
TABLE 298 SOUTH KOREA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 322
TABLE 299 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 323
TABLE 300 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 323
TABLE 301 SOUTH KOREA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 324
TABLE 302 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE, 2023–2030 (USD MILLION) 324
TABLE 303 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES,
BY TYPE, 2023–2030 (USD MILLION) 325
TABLE 304 SOUTH KOREA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 325
TABLE 305 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 326
TABLE 306 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 326
TABLE 307 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 327
TABLE 308 SOUTH KOREA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 327
TABLE 309 SOUTH KOREA: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 328
TABLE 310 AUSTRALIA: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 329
TABLE 311 AUSTRALIA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 329
TABLE 312 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 330
TABLE 313 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 330
TABLE 314 AUSTRALIA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 331
TABLE 315 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 331
TABLE 316 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES,
BY TYPE, 2023–2030 (USD MILLION) 332
TABLE 317 AUSTRALIA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 332
TABLE 318 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 333
TABLE 319 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 333
TABLE 320 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 334
TABLE 321 AUSTRALIA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 334
TABLE 322 AUSTRALIA: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 335
TABLE 323 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 336
TABLE 324 REST OF ASIA PACIFIC: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 336
TABLE 325 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 337
TABLE 326 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 337
TABLE 327 REST OF ASIA PACIFIC: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 338
TABLE 328 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS,
BY TYPE, 2023–2030 (USD MILLION) 338
TABLE 329 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 339
TABLE 330 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 339
TABLE 331 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 340
TABLE 332 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 340
TABLE 333 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 341
TABLE 334 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 341
TABLE 335 REST OF ASIA PACIFIC: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 342
TABLE 336 LATIN AMERICA: KEY MACROECONOMIC INDICATORS 343
TABLE 337 LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY COUNTRY,
2023–2030 (USD MILLION) 343
TABLE 338 LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 343
TABLE 339 LATIN AMERICA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 344
TABLE 340 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 344
TABLE 341 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 345
TABLE 342 LATIN AMERICA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 345
TABLE 343 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE, 2023–2030 (USD MILLION) 346
TABLE 344 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 346
TABLE 345 LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 347
TABLE 346 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 347
TABLE 347 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 347
TABLE 348 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 348
TABLE 349 LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 348
TABLE 350 LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 349
TABLE 351 BRAZIL: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 350
TABLE 352 BRAZIL: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 350
TABLE 353 BRAZIL: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 351
TABLE 354 BRAZIL: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 351
TABLE 355 BRAZIL: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 352
TABLE 356 BRAZIL: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 352
TABLE 357 BRAZIL: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 353
TABLE 358 BRAZIL: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 353
TABLE 359 BRAZIL: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 353
TABLE 360 BRAZIL: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 354
TABLE 361 BRAZIL: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 354
TABLE 362 BRAZIL: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 355
TABLE 363 BRAZIL: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 355
TABLE 364 MEXICO: NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 357
TABLE 365 MEXICO: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 357
TABLE 366 MEXICO: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 357
TABLE 367 MEXICO: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS,
BY TYPE, 2023–2030 (USD MILLION) 358
TABLE 368 MEXICO: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 358
TABLE 369 MEXICO: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE,
2023–2030 (USD MILLION) 359
TABLE 370 MEXICO: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 359
TABLE 371 MEXICO: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 360
TABLE 372 MEXICO: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 360
TABLE 373 MEXICO: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 360
TABLE 374 MEXICO: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE,
2023–2030 (USD MILLION) 361
TABLE 375 MEXICO: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 361
TABLE 376 MEXICO: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 362
TABLE 377 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 363
TABLE 378 REST OF LATIN AMERICA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 363
TABLE 379 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 363
TABLE 380 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 364
TABLE 381 REST OF LATIN AMERICA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 364
TABLE 382 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS,
BY TYPE, 2023–2030 (USD MILLION) 365
TABLE 383 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 365
TABLE 384 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 366
TABLE 385 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 366
TABLE 386 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 366
TABLE 387 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 367
TABLE 388 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 367
TABLE 389 REST OF LATIN AMERICA: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 368
TABLE 390 MIDDLE EAST & AFRICA: KEY MACROECONOMIC INDICATORS 369
TABLE 391 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY REGION,
2023–2030 (USD MILLION) 369
TABLE 392 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 370
TABLE 393 MIDDLE EAST & AFRICA: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 370
TABLE 394 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 370
TABLE 395 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 371
TABLE 396 MIDDLE EAST & AFRICA: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE, 2023–2030 (USD MILLION) 371
TABLE 397 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS,
BY TYPE, 2023–2030 (USD MILLION) 371
TABLE 398 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 372
TABLE 399 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 372
TABLE 400 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 372
TABLE 401 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 373
TABLE 402 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 373
TABLE 403 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 374
TABLE 404 MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 374
TABLE 405 GCC COUNTRIES: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 375
TABLE 406 GCC COUNTRIES: DIAGNOSTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 376
TABLE 407 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 376
TABLE 408 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 376
TABLE 409 GCC COUNTRIES: THERAPEUTIC NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 377
TABLE 410 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE, 2023–2030 (USD MILLION) 377
TABLE 411 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 378
TABLE 412 GCC COUNTRIES: NUCLEAR MEDICINE MARKET, BY APPLICATION,
2023–2030 (USD MILLION) 378
TABLE 413 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 378
TABLE 414 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS,
BY TYPE, 2023–2030 (USD MILLION) 379
TABLE 415 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 379
TABLE 416 GCC COUNTRIES: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 380
TABLE 417 GCC COUNTRIES: NUCLEAR MEDICINE MARKET, BY END USER,
2023–2030 (USD MILLION) 380
TABLE 418 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY TYPE,
2023–2030 (USD MILLION) 381
TABLE 419 REST OF MIDDLE EAST & AFRICA: DIAGNOSTIC NUCLEAR MEDICINE MARKET,
BY TYPE, 2023–2030 (USD MILLION) 381
TABLE 420 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR SPECT RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 382
TABLE 421 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR PET RADIOPHARMACEUTICALS, BY TYPE, 2023–2030 (USD MILLION) 382
TABLE 422 REST OF MIDDLE EAST & AFRICA: THERAPEUTIC NUCLEAR MEDICINE MARKET,
BY TYPE, 2023–2030 (USD MILLION) 382
TABLE 423 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR BETA EMITTERS, BY TYPE, 2023–2030 (USD MILLION) 383
TABLE 424 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR BRACHYTHERAPY ISOTOPES, BY TYPE, 2023–2030 (USD MILLION) 383
TABLE 425 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY APPLICATION, 2023–2030 (USD MILLION) 384
TABLE 426 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR DIAGNOSTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 384
TABLE 427 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR SPECT APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 384
TABLE 428 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR PET APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 385
TABLE 429 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET FOR THERAPEUTIC APPLICATIONS, BY TYPE, 2023–2030 (USD MILLION) 385
TABLE 430 REST OF MIDDLE EAST & AFRICA: NUCLEAR MEDICINE MARKET, BY END USER, 2023–2030 (USD MILLION) 386
TABLE 431 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN NUCLEAR MEDICINE, JANUARY 2022–DECEMBER 2025 388
TABLE 432 NUCLEAR MEDICINE MARKET: DEGREE OF COMPETITION 393
TABLE 433 NUCLEAR MEDICINE MARKET: REGION FOOTPRINT 401
TABLE 434 NUCLEAR MEDICINE MARKET: TYPE FOOTPRINT 402
TABLE 435 NUCLEAR MEDICINE MARKET: APPLICATION FOOTPRINT 403
TABLE 436 NUCLEAR MEDICINE MARKET: DETAILED LIST OF KEY STARTUP/SME PLAYERS 406
TABLE 437 NUCLEAR MEDICINE MARKET: COMPETITIVE BENCHMARKING OF KEY EMERGING PLAYERS/STARTUPS, BY REGION 406
TABLE 438 NUCLEAR MEDICINE MARKET: COMPETITIVE BENCHMARKING OF KEY EMERGING PLAYERS/STARTUPS, BY TYPE 407
TABLE 439 NUCLEAR MEDICINE MARKET: COMPETITIVE BENCHMARKING OF KEY EMERGING PLAYERS/STARTUPS, BY APPLICATION 408
TABLE 440 NUCLEAR MEDICINE MARKET: PRODUCT APPROVALS,
JANUARY 2022– DECEMBER 2025 408
TABLE 441 NUCLEAR MEDICINE MARKET: DEALS, JANUARY 2022–DECEMBER 2025 410
TABLE 442 NUCLEAR MEDICINE MARKET: EXPANSIONS, JANUARY 2022–DECEMBER 2025 412
TABLE 443 NUCLEAR MEDICINE MARKET: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 413
TABLE 444 NOVARTIS AG: COMPANY OVERVIEW 414
TABLE 445 NOVARTIS AG: PRODUCTS OFFERED 415
TABLE 446 NOVARTIS AG: PRODUCT APPROVALS, JANUARY 2022–DECEMBER 2025 416
TABLE 447 NOVARTIS AG: DEALS, JANUARY 2022–DECEMBER 2025 417
TABLE 448 NOVARTIS AG: EXPANSIONS, JANUARY 2022–DECEMBER 2025 418
TABLE 449 LANTHEUS HOLDINGS, INC.: COMPANY OVERVIEW 421
TABLE 450 LANTHEUS HOLDINGS, INC.: PRODUCTS OFFERED 422
TABLE 451 LANTHEUS HOLDINGS, INC.: DEALS, JANUARY 2022–DECEMBER 2025 423
TABLE 452 GE HEALTHCARE: COMPANY OVERVIEW 425
TABLE 453 GE HEALTHCARE: PRODUCTS OFFERED 426
TABLE 454 GE HEALTHCARE: PRODUCT APPROVALS, JANUARY 2022–DECEMBER 2025 427
TABLE 455 GE HEALTHCARE: DEALS, JANUARY 2022–DECEMBER 2025 427
TABLE 456 GE HEALTHCARE: EXPANSIONS, JANUARY 2022–DECEMBER 2025 429
TABLE 457 GE HEALTHCARE: OTHER DEVELOPMENTS, JANUARY 2022–DECEMBER 2025 429
TABLE 458 CURIUM: COMPANY OVERVIEW 431
TABLE 459 CURIUM: PRODUCTS OFFERED 431
TABLE 460 CURIUM: PRODUCT APPROVALS & ENHANCEMENTS,
JANUARY 2022–DECEMBER 2025 435
TABLE 461 CURIUM: DEALS, JANUARY 2022–DECEMBER 2025 436
TABLE 462 CURIUM: EXPANSIONS, JANUARY 2022–DECEMBER 2025 438
TABLE 463 CURIUM: OTHER DEVELOPMENTS, JANUARY 2022–DECEMBER 2025 438
TABLE 464 TELIX PHARMACEUTICALS LIMITED: COMPANY OVERVIEW 440
TABLE 465 TELIX PHARMACEUTICALS LIMITED: PRODUCTS OFFERED 441
TABLE 466 TELIX PHARMACEUTICALS LIMITED: PRODUCT APPROVALS,
JANUARY 2022–DECEMBER 2025 442
TABLE 467 TELIX PHARMACEUTICALS LIMITED: DEALS, JANUARY 2022–DECEMBER 2025 443
TABLE 468 TELIX PHARMACEUTICALS LIMITED: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 444
TABLE 469 SIEMENS HEALTHINEERS: COMPANY OVERVIEW 446
TABLE 470 SIEMENS HEALTHINEERS: PRODUCTS OFFERED 447
TABLE 471 SIEMENS HEALTHINEERS: DEALS, JANUARY 2022–DECEMBER 2025 448
TABLE 472 SIEMENS HEALTHINEERS: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 448
TABLE 473 CHINA ISOTOPE & RADIATION CORPORATION: COMPANY OVERVIEW 449
TABLE 474 CHINA ISOTOPE & RADIATION CORPORATION: PRODUCTS OFFERED 450
TABLE 475 CHINA ISOTOPE & RADIATION CORPORATION: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 451
TABLE 476 BAYER AG: COMPANY OVERVIEW 452
TABLE 477 BAYER AG: PRODUCTS OFFERED 453
TABLE 478 BAYER AG: DEALS, JANUARY 2022–DECEMBER 2025 454
TABLE 479 BRACCO IMAGING S.P.A.: COMPANY OVERVIEW 455
TABLE 480 BRACCO IMAGING S.P.A.: PRODUCTS OFFERED 455
TABLE 481 BRACCO IMAGING S.P.A.: PRODUCT APPROVALS,
JANUARY 2022–DECEMBER 2025 456
TABLE 482 BRACCO IMAGING S.P.A.: DEALS, JANUARY 2022–DECEMBER 2025 456
TABLE 483 BRACCO IMAGING S.P.A.: OTHER DEVELEOPMENTS,
JANUARY 2022–DECEMBER 2025 457
TABLE 484 CARDINAL HEALTH: COMPANY OVERVIEW 458
TABLE 485 CARDINAL HEALTH: PRODUCTS OFFERED 459
TABLE 486 CARDINAL HEALTH: DEALS, JANUARY 2022–DECEMBER 2025 462
TABLE 487 JUBILANT PHARMOVA LIMITED: COMPANY OVERVIEW 463
TABLE 488 JUBILANT PHARMOVA LIMITED: PRODUCTS OFFERED 464
TABLE 489 JUBILANT PHARMOVA LIMITED: PRODUCT APPROVALS,
JANUARY 2022–DECEMBER 2025 466
TABLE 490 JUBILANT PHARMOVA LIMITED: DEALS, JANUARY 2022–DECEMBER 2025 466
TABLE 491 JUBILANT PHARMOVA LIMITED: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 467
TABLE 492 ELI LILLY AND COMPANY: COMPANY OVERVIEW 468
TABLE 493 ELI LILLY AND COMPANY: PRODUCTS OFFERED 469
TABLE 494 ELI LILLY AND COMPANY: DEALS, JANUARY 2022–DECEMBER 2025 470
TABLE 495 ELI LILLY AND COMPANY: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 471
TABLE 496 BWXT MEDICAL LTD.: COMPANY OVERVIEW 472
TABLE 497 BWXT MEDICAL LTD.: PRODUCTS OFFERED 472
TABLE 498 BWXT MEDICAL LTD.: PRODUCT APPROVALS, JANUARY 2022–DECEMBER 2025 473
TABLE 499 NTP RADIOISOTOPES SOC LTD.: COMPANY OVERVIEW 474
TABLE 500 NTP RADIOISOTOPES SOC LTD.: PRODUCTS OFFERED 474
TABLE 501 NTP RADIOISOTOPES SOC LTD.: DEALS, JANUARY 2022–DECEMBER 2025 475
TABLE 502 ECKERT & ZIEGLER: COMPANY OVERVIEW 476
TABLE 503 ECKERT & ZIEGLER: PRODUCTS OFFERED 477
TABLE 504 ECKERT & ZIEGLER: PRODUCT APPROVALS, JANUARY 2022–DECEMBER 2025 478
TABLE 505 ECKERT & ZIEGLER: DEALS, JANUARY 2022–DECEMBER 2025 478
TABLE 506 ECKERT & ZIEGLER: EXPANSIONS, JANUARY 2022–DECEMBER 2025 480
TABLE 507 JOINT STOCK COMPANY ISOTOPE (ISOTOPE JSC): COMPANY OVERVIEW 481
TABLE 508 ISOTOPE JSC: PRODUCTS OFFERED 481
TABLE 509 ISOTOPE JSC: DEALS, JANUARY 2022–DECEMBER 2025 482
TABLE 510 ISOTOPE JSC: OTHER DEVELOPMENTS, JANUARY 2022–DECEMBER 2025 483
TABLE 511 PDRADIOPHARMA INC.: COMPANY OVERVIEW 484
TABLE 512 PDRADIOPHARMA INC.: PRODUCTS OFFERED 484
TABLE 513 PDRADIOPHARMA INC.: PRODUCT LAUNCHES & APPROVALS,
JANUARY 2022–DECEMBER 2025 486
TABLE 514 PDRADIOPHARMA INC.: DEALS, JANUARY 2022–DECEMBER 2025 487
TABLE 515 PDRADIOPHARMA INC.: EXPANSIONS, JANUARY 2022–DECEMBER 2025 487
TABLE 516 ITM ISOTOPE TECHNOLOGIES MUNICH SE: COMPANY OVERVIEW 488
TABLE 517 ITM ISOTOPE TECHNOLOGIES MUNICH SE: PRODUCTS OFFERED 489
TABLE 518 ITM ISOTOPE TECHNOLOGIES MUNICH SE: PRODUCT APPROVALS,
JANUARY 2022–DECEMBER 2025 489
TABLE 519 ITM ISOTOPE TECHNOLOGIES MUNICH SE: DEALS,
JANUARY 2022–DECEMBER 2025 490
TABLE 520 ITM ISOTOPE TECHNOLOGIES MUNICH SE: EXPANSIONS,
JANUARY 2022–DECEMBER 2025 491
TABLE 521 ITM ISOTOPE TECHNOLOGIES MUNICH SE: OTHER DEVELOPMENTS,
JANUARY 2022–DECEMBER 2025 492
TABLE 522 NARODOWE CENTRUM BADAŃ JĄDROWYCH OŚRODEK RADIOIZOTOPÓW – POLATOM: COMPANY OVERVIEW 493
TABLE 523 NARODOWE CENTRUM BADAŃ JĄDROWYCH OŚRODEK RADIOIZOTOPÓW – POLATOM: PRODUCTS OFFERED 494
TABLE 524 NARODOWE CENTRUM BADAŃ JĄDROWYCH OŚRODEK RADIOIZOTOPÓW – POLATOM: DEALS, JANUARY 2022–DECEMBER 2025 496
TABLE 525 ANSTO: COMPANY OVERVIEW 497
TABLE 526 ANSTO: PRODUCTS OFFERED 498
TABLE 527 ANSTO: DEALS, JANUARY 2022–DECEMBER 2025 499
TABLE 528 ANSTO: OTHER DEVELOPMENTS, JANUARY 2022–DECEMBER 2025 499
TABLE 529 RISK ASSESSMENT: NUCLEAR MEDICINE MARKET 523
LIST OF FIGURES
FIGURE 1 NUCLEAR MEDICINE MARKET SEGMENTATION & REGIONAL SCOPE 42
FIGURE 2 MARKET SCENARIO 50
FIGURE 3 GLOBAL NUCLEAR MEDICINE MARKET, 2023–2030 50
FIGURE 4 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN NUCLEAR MEDICINE MARKET, 2022–2025 51
FIGURE 5 DISRUPTIONS INFLUENCING GROWTH OF NUCLEAR MEDICINE MARKET 52
FIGURE 6 HIGH-GROWTH SEGMENTS IN NUCLEAR MEDICINE MARKET, 2025–2030 53
FIGURE 7 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN NUCLEAR MEDICINE MARKET, IN TERMS OF VALUE, DURING FORECAST PERIOD 54
FIGURE 8 RAPID EXPANSION OF RADIOLIGAND AND TARGETED RADIONUCLIDE THERAPY TO DRIVE MARKET GROWTH 55
FIGURE 9 DIAGNOSTIC NUCLEAR MEDICINE MARKET IN NORTH AMERICA ACCOUNTED FOR LARGEST SHARE IN 2024 56
FIGURE 10 INDIA TO REGISTER HIGHEST GROWTH RATE DURING FORECAST PERIOD 56
FIGURE 11 GROWTH RATES OF EMERGING ECONOMIES TO BE HIGHER THAN THOSE OF DEVELOPED ECONOMIES DURING FORECAST PERIOD 57
FIGURE 12 NUCLEAR MEDICINE MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES,
AND CHALLENGES 59
FIGURE 13 ESTIMATED NUMBER OF NEW CASES IN EMERGING ECONOMIES, 2022–2050 70
FIGURE 14 NUCLEAR MEDICINE MARKET: PORTER’S FIVE FORCES ANALYSIS 76
FIGURE 15 GLOBAL GDP GROWTH, 2020–2030 79
FIGURE 16 NUCLEAR MEDICINE MARKET: VALUE CHAIN ANALYSIS (2024) 81
FIGURE 17 NUCLEAR MEDICINE MARKET: ECOSYSTEM ANALYSIS 83
FIGURE 18 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS’ BUSINESSES 91
FIGURE 20 PATENT APPLICATION TRENDS IN NUCLEAR MEDICINE MARKET, 2015–2026 107
FIGURE 21 JURISDICTION & TOP APPLICANT ANALYSIS FOR NUCLEAR MEDICINE MARKET 107
FIGURE 22 TOP APPLICANTS & OWNERS (COMPANIES/INSTITUTIONS) FOR NUCLEAR MEDICINE MARKET (JANUARY 2015–FEBRUARY 2026) 108
FIGURE 23 MARKET POTENTIAL OF AI/GEN AI ON NUCLEAR MEDICINE ACROSS END USERS 113
FIGURE 24 IMPACT OF AI/GEN AI ON INTERCONNECTED & ADJACENT ECOSYSTEMS 116
FIGURE 25 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR END USERS 139
FIGURE 26 KEY BUYING CRITERIA FOR TOP THREE END USERS 141
FIGURE 27 NORTH AMERICA: NUCLEAR MEDICINE MARKET SNAPSHOT 211
FIGURE 28 ASIA PACIFIC: NUCLEAR MEDICINE MARKET SNAPSHOT (2024) 290
FIGURE 29 REVENUE ANALYSIS OF KEY PLAYERS IN NUCLEAR MEDICINE MARKET,
2020–2024 392
FIGURE 30 MARKET SHARE ANALYSIS OF KEY PLAYERS IN NUCLEAR
MEDICINE MARKET, 2024 393
FIGURE 31 NUCLEAR MEDICINE MARKET: BRAND/PRODUCT COMPARATIVE ANALYSIS 396
FIGURE 32 EV/EBITDA OF KEY VENDORS 397
FIGURE 33 YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK
BETA OF KEY VENDORS 397
FIGURE 34 NUCLEAR MEDICINE MARKET: COMPANY EVALUATION MATRIX
(KEY PLAYERS), 2024 399
FIGURE 35 NUCLEAR MEDICINE MARKET: COMPANY FOOTPRINT 400
FIGURE 36 NUCLEAR MEDICINE MARKET: COMPANY EVALUATION MATRIX
(STARTUPS/SMES), 2024 405
FIGURE 37 NOVARTIS AG: COMPANY SNAPSHOT (2024) 415
FIGURE 38 LANTHEUS HOLDINGS, INC.: COMPANY SNAPSHOT (2024) 422
FIGURE 39 GE HEALTHCARE: COMPANY SNAPSHOT (2024) 426
FIGURE 40 TELIX PHARMACEUTICALS LIMITED: COMPANY SNAPSHOT (2024) 441
FIGURE 41 SIEMENS HEALTHINEERS: COMPANY SNAPSHOT (2024) 447
FIGURE 42 CHINA ISOTOPE & RADIATION CORPORATION: COMPANY SNAPSHOT (2024) 450
FIGURE 43 BAYER AG: COMPANY SNAPSHOT (2024) 453
FIGURE 44 CARDINAL HEALTH: COMPANY SNAPSHOT (2024) 459
FIGURE 45 JUBILANT PHARMOVA LIMITED: COMPANY SNAPSHOT (2024) 464
FIGURE 46 ELI LILLY AND COMPANY: COMPANY SNAPSHOT (2024) 469
FIGURE 47 ECKERT & ZIEGLER: COMPANY SNAPSHOT (2024) 477
FIGURE 48 ANSTO: COMPANY SNAPSHOT (2024) 497
FIGURE 49 RESEARCH METHODOLOGY: NUCLEAR MEDICINE MARKET 506
FIGURE 50 RESEARCH DESIGN 507
FIGURE 51 BREAKDOWN OF PRIMARY INTERVIEWS: BY COMPANY TYPE, DESIGNATION,
AND REGION 511
FIGURE 52 RESEARCH METHODOLOGY: HYPOTHESIS BUILDING 513
FIGURE 53 SUPPLY-SIDE MARKET ESTIMATION: REVENUE SHARE ANALYSIS 515
FIGURE 54 NUCLEAR MEDICINE MARKET: REVENUE SHARE ANALYSIS ILLUSTRATION 515
FIGURE 55 NUCLEAR MEDICINE MARKET: REVENUE ANALYSIS OF TOP FOUR COMPANIES (2024) 516
FIGURE 56 NUCLEAR MEDICINE MARKET SIZE ESTIMATION: BOTTOM-UP APPROACH 517
FIGURE 57 NUCLEAR MEDICINE MARKET SIZE ESTIMATION: TOP-DOWN APPROACH 518
FIGURE 58 CAGR PROJECTIONS FROM ANALYSIS OF MARKET DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES IN NUCLEAR MEDICINE INDUSTRY 519
FIGURE 59 CAGR PROJECTIONS: NUCLEAR MEDICINE MARKET, 2025−2030 520
FIGURE 60 DATA TRIANGULATION METHODOLOGY 521